Posterior Cervical Arthrodeses: Occiput-C2 and C1-C2
Jaime A. Gomez
Daniel J. Hedequist
DEFINITION
The occipitocervical articulation is formed by the occiput, the atlas (C1), and the axis (C2). This functional unit provides a large degree of mobility and range of motion through the strong ligamentous structures and cup-shaped joints.
Over 50% of the total axial rotation occurs between C1 and C2, whereas flexion-extension movement predominantly occurs at the occipitoatlantal junction which allows approximately 20 degrees of extension.1
Cervical spine range of motion is significantly decreased in children after posterior occipitocervical arthrodesis. Axial rotation is the most affected, decreasing 30 degrees in each direction, flexion and extension each decrease by 13 degrees, and lateral bending by 7 degrees in each direction.25
Excessive movement at this junction due to either bony or ligamentous abnormalities causes instability. A wide variety of pathologies such as genetic and congenital developmental abnormalities, trauma, tumors, and inflammatory and degenerative conditions can lead to upper cervical spine instability.
Depending on the degree of displacement and spinal canal compromise, cord compression and myelopathy may occur.
Major instability is usually addressed with surgical occipitocervical or C1-C2 arthrodesis. Since Foerster first described a technique for occipitocervical arthrodesis using fibular strut graft in 1927, several procedures have been reported with variable rates of fusion and techniques of stabilization.
In this chapter, brief information on upper cervical spine instability is given and general principles of occipitocervical and C1-C2 arthrodesis are discussed. Also, different techniques developed for posterior occipitocervical fusion are described in detail.
Modern instrumentation techniques have dramatically changed the instrumentation potential and wiring strategies are being replaced for screw instrumentation techniques. These techniques can decrease the risk of cord damage from wire passage, increase biomechanically fixation, and improves fusion rates.13, 16
ANATOMY
It is important to understand that the pediatric upper cervical spine is not a “miniature model” of the adult spine. The cervical spine approaches adult size and shape by ages 8 to 12 years, as growth cartilage fuses and vertebral bodies gradually lose their oval or wedge shape and become more rectangular.
The upper cervical spine has unique development, anatomy, and biomechanics.
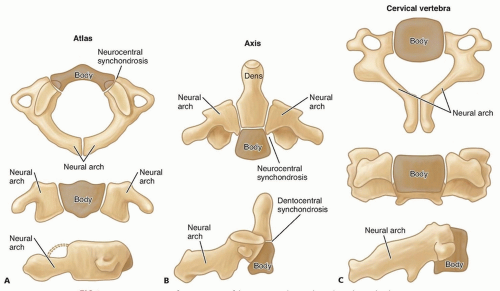
The C1 develops from three ossification centers, a body and two neurocentral arches, which become visible by age 1 year (FIG 1A).
The neurocentral synchondroses fuse with the body at about 7 years of age and may be mistaken for fractures on radiographs.18
The C2 is derived from five primary ossification centers: the two neural arches or lateral masses, the two halves of the dens, and the body.
There are two secondary centers: the ossiculum terminale and the inferior ring apophysis (FIG 1B).
The two halves of the odontoid are generally fused at birth but may persist as two centers, known as the dens bicornis.19
The dentocentral synchondrosis, which separates the dens from the body, closes between the ages of 5 and 7 years (FIG 1B). Until the ossification is complete, it gives the appearance of a “cork in a bottle” on an open mouth odontoid view.
The tip of the dens appears at age 3 years and is fused by age 12 years.
After skeletal maturity, the C1 does not have a body as such and is shaped like a ring. The flat, cup-shaped articular surface under the occipital condyle allows for flexion, extension, and some bending. The dens articulates with C1 through the dorsal facet of the anterior arch. C1-C2 articulation allows for rotation.1 The vertebral artery passes through the foramen that is located in the transverse processes.
The ligamentous structure allows for a wide range of motion of the upper cervical spine while maintaining stability. The short ligaments at the base of the skull are as follows (FIG 1C):
The tectorial membrane anteriorly from the foramen magnum is a continuation of the posterior longitudinal ligament that provides considerable support.
Cruciate ligament, which includes transfer ligaments, restrains against atlantoaxial anteroposterior (AP) translation.
Alar and apical ligaments, which run from foramen magnum to the odontoid, act as secondary stabilizers.
Posterior atlanto-occipital ligaments are the continuation of the ligamentum flavum.
Vertebral artery anatomy can also be different in children compared to adults. Younger patients have vertebral arteries that are significantly closer to the midline over the superior border of C1 than older patients. Ninety-seven percent of vertebral arteries are greater than a centimeter lateral to the midline.12
PATHOGENESIS
Fundamentally, upper cervical spine instability can develop from osseous or ligamentous abnormalities resulting from acquired or congenital disorders. As a result of instability, excessive motion and spinal cord compression may occur at the occipitoatlantal and/or atlantoaxial joint.
In nontraumatic conditions, ligamentous laxity (particularly in the transverse ligament) or abnormalities of the odontoid cause instability.
In Grisel syndrome, a type of atlantoaxial rotatory displacement, inflammation of the retropharyngeal space, caused by upper respiratory tract infections or by adenotonsillectomy, spreads through the pharyngovertebral veins to the ligaments of the upper cervical spine. This results in impairment of the transverse atlantal ligament and instability.27
In Down syndrome, the main cause of atlantoaxial instability is the laxity of the transverse ligament, which holds the dens against the posterior border of the anterior arch. Also, malformation of the odontoid can be observed in this condition.8
Klippel-Feil syndrome is associated with congenital cervical anomalies, such as occipitocervical synostosis, basilar impression, and anomalies of the odontoid, and can be associated with instability, stenosis, or both.
Odontoid anomalies include aplasia, hypoplasia, duplication, third condyle, persisting ossiculum terminale, and os odontoideum.
NATURAL HISTORY
Patients with upper cervical instability frequently have other associated pathologic conditions in the occipitocervical region such as spinal stenosis, basilar impression, cervical fusions, occipitalization, or congenital anomalies of the C1 or C2 (dens), and central nervous system abnormalities.
When one encounters one of these conditions, others should be sought also.
Instability of the upper cervical spine and stenosis often are two major factors in the development of myelopathy.
Patients who are symptomatic at initial presentation are often at risk for progressive neurologic symptoms. Once cervical myelopathy develops, it rarely resolves entirely.
Paralysis and death are rare but may be encountered in patients with upper cervical spine instability.
PATIENT HISTORY AND PHYSICAL FINDINGS
Upper cervical spine instability is rare in patients without predisposing conditions or trauma.
The instability is usually determined in radiographic examination of the children with syndromes or conditions known to have frequent involvement of the musculoskeletal system.24 An orthopaedic surgeon is usually consulted for children with such conditions.
Clinical presentation can vary because of the associated syndromes and anomalies.
Patients may present with symptoms such as loss of range of motion, stiffness, mechanical pain of the head or neck, and torticollis.
It is not uncommon to see patients presenting with neurologic symptoms, which can vary from minor sensory or motor disturbances to established myelopathy. Neurologic symptoms or signs result from mechanical compression of the spinal cord or nerve roots.
Torticollis may be the presenting symptom of rotatory or postinfectious atlantoaxial instability.
According to the degree of compression and the affected site of the spinal cord, signs and symptoms can vary. They may include loss of physical endurance, difficulty walking, weakness, and upper motor neuron signs (spasticity, hyperreflexia, clonus, Babinski sign), which can be seen with anterior spinal column involvement.
Pain deficits and proprioception and vibratory sense deficits can be seen with posterior spinal column involvement.
Increased nasal resonance may also be observed. It may occur because of the decreased size of the nasopharynx resulting from anterior displacement of the C1.
Vertebral artery distortion and insufficiency may lead to bizarre symptoms such as syncopal episodes, sudden postural collapse without unconsciousness, change in behavior, dizziness, and seizures.
In cerebellar involvement, nystagmus, ataxia, and incoordination are the common findings.
Neurogenic bladder and bowel, cranial nerve involvement, paraplegia, hemiplegia, and quadriplegia should be kept in mind; sometimes, the patient presents with only one of these findings.
IMAGING AND OTHER DIAGNOSTIC STUDIES
Standard radiographs include AP, open mouth odontoid, and lateral (neutral and flexion-extension) cervical spine views.
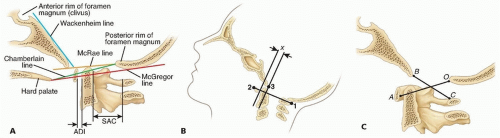
Instability can be identified on the lateral flexion-extension view. Atlantoaxial instability is diagnosed on the basis of an increased atlantodental interval (ADI).
The ADI is measured from the anterior aspect of the dens to the posterior aspect of the anterior ring of the C1 (FIG 2A).
In children older than 8 years and in adults, the ADI should be 3 mm or less, whereas in younger children, the ADI should be 4 mm or less (some consider 4.5 to 5 mm acceptable).14
In children, we consider an ADI of 4 mm or more as evidence of atlantoaxial instability. This measurement does not always correlate with the degree of brainstem or cord compression (as seen on magnetic resonance imaging [MRI]), however. An asymptomatic patient may have instability.
Space available for the spinal cord (SAC) is measured from the posterior border of the dens to the anterior border of the posterior tubercle. According to Steel’s rule of thirds,22 SAC should be about one-third of the diameter of the ring of C1 (see FIG 2A).
This safe zone allows for some degree of pathologic displacement. Displacement of more than one-third of the diameter causes cord compression.
This measurement directly describes the SAC, which is highly associated with the neurologic involvement.
The relationship between the foramen magnum, C1, and odontoid can be determined in lateral radiographs.
The line of McRae connects the anterior rim of the foramen magnum to the posterior rim (see FIG 2A).
The upper tip of the odontoid should normally be 1 cm below the anterior margin of the foramen magnum.
If the effective sagittal diameter of the canal (length of the line) is less than 19 mm, neurologic symptoms occur.
The line of Chamberlain is drawn from the posterior margin of the hard palate to the posterior margin of the foramen magnum (see FIG 2).
The tip of the odontoid should be 6 mm below this line. It bisects the line in basilar invagination. However, determination of the landmarks can be difficult on plain radiographs.
The McGregor line is drawn from the most caudal point of the occipital projection to the posterior edge of the hard plate (see FIG 2A).
This line is one of the best for detecting basilar impression because the osseous landmarks can usually be seen at all ages. If the tip of the odontoid process lies more than 4.5 mm above this line, the finding is consistent with basilar impression.
The line of Wackenheim is drawn parallel to the posterior surface of the clivus (see FIG 2A).
The inferior extension of the line should be in touch with the posterior tip of the odontoid. In basilar invagination, it is over that line.
The Wiesel-Rothman line is drawn connecting the anterior and posterior arches of the C1. Two lines are drawn perpendicular to this line, one through the basion and the
other through the posterior margin of the anterior arch of the C1 (FIG 2B).
A change in the distance (x) between these lines of more than 1 mm in flexion and extension indicates increased abnormal translational motion.
The ratio of Power is calculated from a line drawn from the basion to the posterior arch of the C1 and a second line from the opisthion to the anterior arch of the C1 (FIG 2C). The length of the first line is divided by the length of the second.
A ratio of less than 1.0 is normal.
A ratio of 1.0 or more is abnormal and is diagnostic of anterior occipitoatlantal dislocation.
MRI is useful to identify pathologic changes at the dura mater and spinal cord as well as additional soft tissue pathologies.
Functional MRI scans performed in flexion and extension can be used to assess dynamic brainstem or cord compression.
CT scan is essential for screw trajectory planning and it can also provide additional information regarding the bony anomalies.
Two-dimensional (2-D) and three-dimensional (3-D) reconstructions can clarify the course of the vertebral artery and careful attention should be paid to the location of the artery through the foramen transversarium of C2, where is most likely to be injured.
In atlantoaxial rotational displacement, pathoanatomy is determined by fine-cut dynamic CT scan with left-right rotation of the head.
CT or magnetic resonance (MR) angiogram can be useful to evaluate the vertebral artery anatomy prior to instrumentation of C1-C2.
DIFFERENTIAL DIAGNOSIS
Pseudosubluxation
Os odontoideum
Congenital muscular torticollis
Ankylosing spondylitis
NONOPERATIVE MANAGEMENT
Children with known risk of upper cervical instability should be evaluated carefully. Especially patients with congenital syndromes associated with upper cervical spine instability should have periodic clinical and radiographic examinations until maturity.24
Upper cervical spine radiographs including AP, lateral (neutral and flexion-extension views), and open mouth odontoid views are obtained periodically to assess and detect any trends and changes.
Patients and parents should be educated about the diagnosis and natural history of the disorder and encouraged to report any symptoms as soon as they occur.
Because of bone and ligament abnormalities, patients with upper cervical spine instability have a greater risk of spinal cord injury even with minor trauma and even when they are asymptomatic.
As previously described, periodic observation should be done, and if any progression is noticed, the patient should be prepared for appropriate surgical stabilization when indicated.
In children, we consider an ADI of 4 mm or more as evidence of atlantoaxial instability. Documented significant instability at the atlanto-occipital or atlantoaxial joints is an indication for posterior arthrodesis of occiput-C2 and C1-C2 arthrodesis, respectively.
In some congenital disorders such as Morquio syndrome, progression of the instability is frequent; in these cases, prophylactic fusion should be considered before neurologic symptoms occur.24
However, in Down syndrome, the patients with instability are usually asymptomatic, and in most cases, signs and symptoms progress slowly. Restriction of high-risk activities usually is appropriate. If the clinical symptoms persist or neurologic symptoms are starting to occur in the setting of significant instability, surgical treatment is indicated.8, 21
Children with congenital fusion of cervical vertebrae (mostly in Klippel-Feil syndrome) should be restricted from high-risk sports. Patients with progressive symptomatic segmental instability or neurologic compromise are candidates for surgical stabilization.23
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree