Dupuytren disease (DD) is a benign, generally painless, fibroproliferative disorder affecting the palmar fascia that often leads to progressive contractures of the fingers and thumb (FIG 1).
These contractures can be severe having a significant impact on hand function.
Although most estimates give the incidence of DD as 3% to 6% of Caucasians, some studies have reported rates as high as 42%.46 This would mean that as many as 13.5 to 27 million people in the United States and Europe could be affected.
For a detailed description of normal and pathologic anatomy, please see the excellent description in Dr. Rayan’s chapter 143, Surgical Treatment of Dupuytren’s Disease.
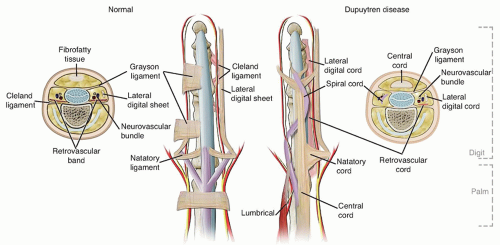
A particular concern is the location of the digital nerve when there is a spiral cord. The presence of a spiral nerve can be predicted if the patient has a soft, fatty mass lying between the distal palmar flexion crease and the proximal finger flexion crease. In 44 dissections, if a patient had this mass, there was a spiral nerve 90% of the time.60
The neurovascular bundle usually makes a straight line in the digit. When the pretendinous band, the spiral band, and the lateral digital sheet become involved in DD, they contract and become straight displacing the neurovascular bundle centrally and palmarly (FIG 2).44
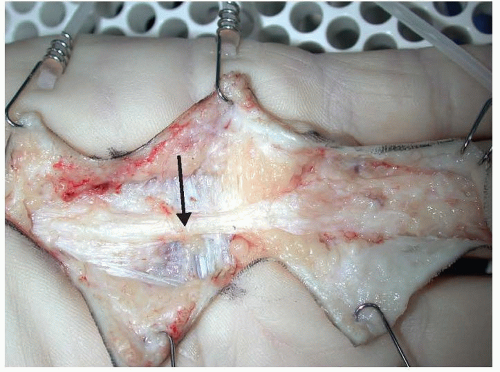
FIG 1 • Surgical dissection showing a pretendinous Dupuytren cord (arrow) affecting the palm and finger.
When the spiral cord contracts, it causes the neurovascular bundle and fibrofatty tissue to become more superficial. This causes redundant skin and fibrofatty tissue to become a soft, pliable mass in the palm that measures approximately 2 cm in diameter.
The spiral nerve occurs most commonly in the ring and small fingers when there is a proximal interphalangeal (PIP) joint contracture of 30 degrees.60
DD is believed to be inherited as an autosomal dominant trait with incomplete penetrance. Hu et al32 was able to identify an autosomal dominant gene for DD on chromosome 16q in a Swedish family.
The progression of DD is not uniform. Cords can be nodular or nonnodular based on the number of localized cellular nodules in a cord. Nodular cords are hypercellular, the majority of which are alpha smooth muscle actin (α-SMA) positive cells. Nonnodular cords are the most contracted. Cells in nodular cords are responsible for cord contraction. Stress shielding leads to myofibroblast apoptosis that results in cords becoming less cellular.69
Normal palmar fascia is composed largely of type I collagen. DD cords are largely composed of type III collagen.10
Type I (proliferative stage): All lesions show mitotic figures. Mitotic figures are regular in shape and are rare. Cells have round nuclei.
Type II (fibrocellular stage): There is high cellularity but no mitotic figures. Cell nuclei are elongated with abundant wavy collagen fibers. Silver stain shows a dense reticulin network in the more cellular areas.
Type III (fibrotic stage): There are dense fibrous cords that rarely contain cells with elongated nuclei.
The highest incidence of recurrence is in patients with the type I histologic group at 54%. By 8 to 9 years postoperatively, the recurrence rate increases to 71%.
The major source of contraction in DD is the myofibroblast. This cell can generate significant contractile forces and has characteristics of both smooth muscle and fibroblasts.5,23
Prostaglandin F2α stimulates significant contracture of myofibroblasts, whereas prostaglandin E2 causes relaxation.5,35
DD tissue contains large amounts of transforming growth factor beta (TGF-β1 and TGF-β2) that has a significant effect on myofibroblast proliferation.6
TGF-β1 is a mechanotransduction cytokine that causes increased contractile force of myofibroblasts during the early phase of attachment and contractions as well as an increased contractile response to mechanical stimuli.9
A Z19 transcription factor gene increases the potency of TGF-β1.
Stage I: the proliferative or nodular stage. In this stage, patients have a nodule or nodules within the palmar fascia. The nodules are predominantly cellular, composed of peripheral, perivascular spindle-shaped hyperplastic fibroblasts with irregular hyperchromatic nuclei. The nodules tend to be vascular with reactive tissue around the periphery. There is no increase in collagen deposition. The hyperplastic cells disrupt the continuity of the normal palmar fascia.
Table 1 Selected Genes and Proteins Upregulated in Dupuytren Disease
A disintegrin and metalloproteinase domain (ADAM) 12
Alpha smooth muscle actin (α-SMA)
β-1 integrin
Cadherin 11 (CDH11)
Collagen I, collagen V, collagen VIII
Contactin 1 (CNTN1)
Fibronectin
Heat shock protein 47 (HSP47)
Laminin
Leucine-rich repeat (LRR) domain-containing 17
V-maf musculoaponeurotic fibrosarcoma oncogene homolog B (MafB)
Periostin, osteoblast specific factor (POSTN)
Postsynaptic density protein-95 (PSD-95)
Tenascin C
Tissue inhibitor of metalloproteinase (TIMP-1)
Transforming growth factor-β2 (TGF-β2)
Zonula occludens-1 protein (ZO-1)
From Black EM, Blazar PE. Dupuytren disease: an evolving understanding of an age-old disease. J Am Acad Orthop Surg 2011;19(12):746-757.
Stage II: the involutional or active disease stage. In this stage, there is nodular thickening of the palmar fascia with the beginning of joint contracture. Fibroblasts align themselves along lines of stress. They become more mature and decrease in size and number. The predominate cell type is myofibroblast. There are definite cords composed of well-aligned, mature collagen fibers and few scattered cells.
Stage III: the residual or advanced disease stage. In this stage, there are diffuse, thick fibrotic cords that become more contracted. The cords are predominantly collagen with few cells that are elongated and compressed by the collagen fibers. Cell types are both fibroblasts and myofibroblasts.
Table 2 Selected Genes and Proteins Downregulated in Dupuytren Disease
Chitinase 3-like protein 2
Collagen XV
Cornea-derived transcript 6 (CDT6)
Cysteine dioxygenase 1 (CDO1)
Matrix metallopeptidase 27 (MMP27)
Matrix metalloproteinase-3 (MMP3)
Superoxide dismutase (SOD)
Superoxide dismutase 2 (SOD2)
From Black EM, Blazar PE. Dupuytren disease: an evolving understanding of an age-old disease. J Am Acad Orthop Surg 2011;19(12):746-757.
The progression of DD is unpredictable. Nodules may lay dormant for years without progression or can progress rapidly over a matter of months.42 Reilly et al57 reported on 59 patients with Dupuytren nodules. Thirty of the 59 patients had developed a cord at an average of 8.7 years (range 6 to 15 years). However, by 8.7 years only, 5 patients met the criteria for surgery and 7 patients had regressed.57
DD predominantly affects Caucasian males of northern European descent. It has been reported in all races, however.
The peak incidence occurs in the 50s for men and the 60s for women. The literature reports that it is more common in men than women with ratios ranging from 7.5:1 to 5.4:1.42 Other studies report ratios ranging from 9.5:1 to 3:1. These studies have largely looked at populations in Europe and Australia, however. In a more recent study, Anthony et al1 looked at the incidence in the U.S. population and found the male-to-female ratio to be 1.7:1 and noted with advancing age, the ratio approached 1:1.
DD interferes with activities of daily living and work including hair brushing, face washing, holding hands, putting hands in pockets, holding tools, and wearing gloves.
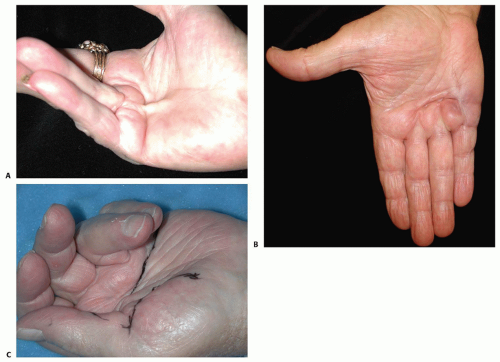
FIG 3 • A. Dupuytren cord affecting the ring finger. B. Pitting and dimpling associated with Dupuytren contracture. C. Multiple Dupuytren cords involving all fingers and the thumb.
DD is associated with a number of medical conditions including diabetes mellitus,3,13 HIV11 frozen shoulder, a high lipid profile,29 and epilepsy.2
It has also been associated with lifestyle risk factors including smoking, alcoholism,12,24,25 manual labor,41 hand and wrist trauma,40 and the use of vibratory tools.64
DD usually begins as painless nodules in the palm. The most common finger involved is the ring finger (60.7%), followed by the small finger (51%), the middle finger (22.5%), the thumb (7.0%), and the index finger (5.8%). The thumb web can also be involved resulting in an adduction contracture (FIG 3A).55
Early in the disease, patients may note thickening of the skin with pitting or dimpling. This may progress to cords that can ultimately lead to contracture of the metacarpophalangeal (MP) and PIP joints (FIG 3B).
DD usually begins with one finger but often progresses to others (FIG 3C).
DD can also occur in ectopic locations such as over the dorsum of the PIP joints (Garrod nodes), in the feet (Lederhose disease), and in the penis (Peyronie disease).30
Five factors have been identified that indicate a DD diathesis. They include onset younger than age 50 years, bilateral involvement, ectopic lesions, male gender, and a positive family history.30,33
Patients with a DD diathesis have a more aggressive disease with higher rates of recurrence.
The “fasciodesis” maneuver is useful for predicting minimum gain in PIP motion after percutaneous needle fasciotomy (PNF).
Flex the MP joint to 90 degrees.
Measure the extension gain at the PIP joint.20
In most instances, radiographic imaging is not necessary to adequately evaluate DD. The only time one might consider imaging is if there is a concern for some other pathology such as significant osteoarthritis or neoplasm. If these are suspected, then appropriate imaging studies could be ordered.
The differential diagnosis of DD is well covered in the chapter on Surgical Treatment of Dupuytren’s Disease.
In the past, a number of nonoperative treatments for DD have met with limited success. These include physical therapy, splinting, dimethylsulfoxide injections, topical vitamins A and E, gamma interferon injections, radiation, and the calcium channel blockers such as nifedipine and verapamil.56
In 1971, Hueston33 performed what he referred to as enzymatic fasciotomy. He injected patients with a mixture of trypsin, hyaluronidase, and lidocaine, followed by a forcible extension maneuver. He was able to obtain full extension of all fingers 15 minutes following injection.33
Table 3 Tubiana Grades of Dupuytren Disease Based on Total Passive Extension Deficit
Grade I
0-45 degrees
Grade II
45-90 degrees
Grade III
90-135 degrees
Grade IV
≥135 degrees
When McCarthy43 studied the long-term results, however, he found that seven of nine patients had developed recurrence within 2 to 3 years.
Injection of triamcinolone acetonide directly into the DD nodules resulted in regression in 97% of patients. The average number of injections was 3.2. There was a 50% recurrence rate between 1 and 3 years after the last injection, however.38
In 2009, Hurst et al34 reported the results of the multicenter collagenase option for reduction of Dupuytren (CORD) I study. This was a double blind, placebo-controlled study in which they injected Dupuytren cords with 0.58 mg collagenase Clostridium histolyticum (CCH) (Xiaflex).34
Xiaflex is a mixture of two synergistic collagenases that tend to rapidly degrade type I and type III collagens.61
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree