Günter Köhler, Katja Evert, Marek Zygmunt and Matthias Evert
4Low-grade endometrial stromal sarcoma
4.1Uterine low-grade endometrial stromal sarcoma
4.1.1General, epidemiology, etiology, pathogenesis, staging
LG-ESS is a pure homologous sarcoma. Endometrial stromal sarcomas had originally been divided into LG-ESS and HG-ESS. This differentiation was primarily based on the number of M/10 HPF. Tumors with < 10M/10HPF were categorized as LG-ESS, while such with ≥ 10M/10HPFwere termed HG-ESS. It had,however, already been known for some time that prognosis did not always correlate with mitotic count (69).Accordingly, the categorization of tumors into LG- and HG-ESS along the lines of MI was abandoned some years ago. Instead, stromal sarcomas were pathologico-anatomically differentiated into endometrial stromal sarcomas (ESS) and undifferentiated endometrial sarcomas (UES) on the basis of cytologic pleomorphism, the degree of nuclear atypia and the presence of TCN. These two entities also differed in terms of their immunologic and cytogenetic profiles (98), a difference that could be drawn on in cases of uncertainty when having to discern between the two.Under this classification, ESS (optionally ESS low-grade) constituted the more differentiated and prognostically more favorable tumor, while UES was comparably less differentiated and clearly the more aggressive of the two. According to the criteria of the time, tumors with an MI of ≥ 10M/10HPF could be ESS, and tumors with an MI < 10M/10HPF could be classified as UES. Differentiation between the two was thus guided by cytologic/nuclear properties and features.
Already at the time, the change in classification and the often still lacking differentiation between the two tumor entities served to compromise the validity or weight of numerous studies.
The current WHO Classification of 2014 (172) has returned to the distinction into LG-ESS and HG-ESS, but now also includes UUS. Tumors previously categorized as UES need to be stratified into HG-ESS with an intermediate but nonetheless poor prognosis on the one hand, and UUS with the worst prognosis on the other.
While not many data have been gathered from randomized studies thus far, largerscale retrospective SEER analyses have been conducted that provide a lot of meaningful information. The current NCCN Guidelines –Uterine Neoplasms (160) are definitely also a helpful resource, notwithstanding the fact that they are almost entirely based on “lower-level evidence” due to the lack of available experience, research and investigation.
According to DKSM data, in Germany, LG-ESS account for 65% of all sarcomas that originate from the endometrial stroma, with UUS and HG-ESS accounting for the remainder. They account for 18.1% of all uterine sarcomas when CS are included, and 26.4% when CS are excluded (115). In a Norwegian cohort of similar size, they accounted for 20% of all uterine sarcomas excluding CS (2). They have an incidence of 0.23 per 100,000 women in southern European countries (146), and about 0.3 per 100,000 women in northern Europe, where it is at its highest in women aged between 45 and 49 years. Incidence increases most steeply during the fourth decade of life, and remains largely stable/constant during and as of the fifth decade (117).
According to various sources, the age of affected women ranges from15 to 96 years. Median age is 42–52 years; 50 to 88% of LG-ESS patients are premenopausal (18, 36, 237). The age range in Germany is 18–80 years according to DKSM data (115), median age is 46 years, and only 18% of presenting women are postmenopausal. LG-ESS thus predominantly arise in young to middle-aged women. Disease in women aged 20 to 30 is by no means the exception. Correspondingly, LG-ESS occur at a considerably younger age than HG-ESS and UUS do (see also Tab. 6.1.1) (36, 115, 128, 199, 207). Black women are said to be affected more frequently than white women (140).
A look at age distribution reveals an epidemiologic correlation between LG-ESS and ESN – the latter arise in patients aged between 23 and 86 years, and 75% of patients are premenopausal (241). LG-ESS and ESN also share clear pathogenetic commonalities. On the one hand, the microscopic differences between these two tumor types are gradual rather than abrupt or clear-cut. On the other hand, both LG-ESS and ESN exhibit t(7;17) translocations and consecutive fusions of the JAZF1 and SUZ12 (previously: JJAZ1) genes as well as t(6;7) translocations (JAZF1/PHF1 gene fusion) (119, 166). Interestingly, this translocation of the former can also be observed in ESN and LG-ESS that exhibit focal smooth muscle differentiation, which allows the conclusion that such tumors originate from a pluripotential mesenchymal precursor cell (progenitor cell) (166, 171). The putative localization of such a progenitor cell within the myometrium could help to explain why LG-ESS and ESN both predominantly arise there. But according to recent research, not all LG-ESS exhibit LG-ESS related gene fusions (see also subchapter DD) (151). Both LG-ESS and ESN express common immunohistochemicalmarkers (CD10, SMA,WT-1), another factor that supports the notion that stromal and myogenic tumors might derive from the same precursor cell (220). Both LG-ESS and ESN are usually DNA diploid, cytogenetically speaking (26).
Regarding epidemiology, it is interesting to note that, compared to LG-ESS, ESN are only rarely described. In the DKSM database, LG-ESS and ESN have a ratio of only 100 to 3 (115). Subtle analyses have revealed that, when ESN and LG-ESS are summed together, the latter account for a substantially larger share (84%) than the former (16%) (218). Given the fact that microscopically differentiating between LG-ESS and ESN is very difficult, this can essentially only be explained by the fact that a nonnegligible number of ESN (for example such with limited infiltration, cf. Chapter 3) are probably classified or diagnosed as LG-ESS just to be “on the safe side” (19). This could also serve to explain why so many LG-ESS take very good courses. The situation here might well be similar to the one involving STUMP and LMS (cf. Chapters 1 and 2).
There is an assumed epidemiologic association between these neoplasms and hyperestrogenic states, including gestation, endometriosis, assisted reproduction and the administration of xenoestrogens. Xenoestrogens have actually been found to be present within tumors themselves. Aromatase expression has also been found in LG-ESS tissues (187). Furthermore, it is also known that variations in the CYP19 gene can promote estrogen production via aromatases. The sum of all data including the expression of ER and PGR give strength to the assumption that estrogens are involved in the etiology, pathogenesis and progression of LG-ESS (186, 187, 188, 189, 190, 191). Average parity is 1.9, and 22% of women have given birth to < 2 children (199, 237).
It is also highly probable that LG-ESS can develop directly from an endometriosis, not least because LG-ESS are often associated with the latter (56, 148) and because aromatase can also be present in endometriotic plaque (219). However, since LG-ESS are also found in extragenital endometriosis, this observation also allows the conclusion to be drawn that the precursor cells are also situated in the endometrial stroma, and not only in the myometrium.
The role of HRT is highly debated, at least regarding epidemiology. In a larger Finnish study, exposure to HRT for longer than five years was accompanied by significant risk elevations for all uterine sarcomas (97). More recent extensive northern European data sources, in contrast, suggest that HRT has no impact on the incidence of stromal sarcomas (117). In general, anamnestic or ongoing tamoxifen or toremifen intake is regarded as an (albeit rare) etiologic factor for all uterine sarcomas that should not be underestimated (16, 87, 89, 105). The pathogenetic mechanism at play is said to be a mutagenic effect rather than the inherent estrogeneous effect, not least because the prognostically unfavorable non-endometrial ER negative EC are almost twice as frequent compared to the normal population when patients have previously been subjected to or are still receiving tamoxifen treatment (161). Such tamoxifen association also applies to CS, HG-ESS, UUS, LMS and heterologous sarcomas (16). At least for CS, experimental studies have shown that the tumorigenesis provoked by tamoxifen is not dictated by the ER (9). Accordingly, it can occasionally so happen that EC and LG-ESS occur simultaneously following tamoxifen treatment (35) and thus constitute the preconditions for the development of CS via collision, at least theoretically.
A medical history of pelvic irradiation is also said to constitute a risk factor for LG-ESS (240).
Since 2009, new FIGO staging and UICC TNM classification have been in place for LMS and stromal sarcomas (76, 182, 214) (see also Tab. 2.1.1). All pelvic and paraaortic LN are defined as regional LN. Simultaneous LG-ESS of the uterine corpus and the ovaries or the pelvis in association with ovarian or pelvic endometriosis should be classified as independent primary tumors.
One problem lies in the fact that the results from numerous studies cannot be simply transferred or said to apply to the new staging. For example, in the old staging system, stage II included such tumors that are confined to the uterus but extend to the cervix. The current stage II, by contrast, refers to disease that has spread beyond the uterus yet only to the pelvis. Stage III has experienced a dramatic adaptation. Tumors that now belong to stage III had previously been categorized as stage IV. Therapeutic recommendations that had in the past applied to stages II and III can thus not simply be adopted for current FIGO and UICC stage II and III. In the monograph at hand, the old data have been adapted to the new classification where necessary.
Low-grade endometrial stromal sarcomas are pure homologous uterine sarcomas that predominantly arise in premenopausal patients. They need to be clearly distinguished and delineated from high-grade endometrial stromal sarcomas and undifferentiated uterine sarcomas. It is very likely that endometrial stromal nodules are a precursor for low-grade endometrial stromal sarcoma. The JAZF1-SUZ12 gene fusion is the most important genetic criterion of both low-grade endometrial stromal sarcomas and stromal nodules. Tamoxifen exposure plays a role in terms of etiology. The impact of hormone replacement therapy on tumorigenesis is subject to debate. New FIGO staging and UICC TNM classification for leiomyosarcomas and stromal sarcomas have been in place since 2009.
4.1.2Macroscopic and microscopic features
LG-ESS predominantly have the appearance of an intramural nodulated tumor with strong similarities to an LM or the rare ESN (Fig. 4.1.1 (A)). Submucously localized tumors can also be pedunculated, prolapse/protrude from the cervix upon further growth (Fig. 4.1.1 (B)–(E)) and thus appear to resemble, or mimic, pedunculated LM or polyps.
Accordingly, between 69 and 75% of LG-ESS are subjected to primary surgery under a diagnosis of LM (18, 115, 199, 237). LG-ESS have a median size of 5.5–5.8 cm (82, 115). Compared to LM, LG-ESS almost always have a softer consistency, but they are considerably more solid than common fibroglandular mucosal polyps. However, this feature is often only discovered during surgery. Larger tumors can entirely displace the uterine cavity (Fig. 4.1.1 (B)). Like LM, the cut surface may exhibit whorled structures, but is usually significantly smoother (Fig. 4.1.1 (A), (B)). Like ESN, the opened tumor often has a yellowish to yolk-yellow (Fig. 4.1.2 (A)), tan-yellow and sometimes light reddish appearance. On gross examination, intramural tumors can appear well-circumscribed and can displace the surrounding myometrium and endometrium. Compressed tumor and surrounding tissue can form a kind of pseudo-capsule (Fig. 4.1.2 (A)). Analogous to the results from microscopy, infiltrations into the musculature are sometimes already visible to the naked eye as cords, small nodules or trabeculae. Macroscopically visible finger-, worm- or tongue-like invasion, when present, are a typical gross characteristic. The tumor margins are more or less smooth, diffuse or clearly infiltrative, depending on the extent of the tumor’s infiltrative growth.
Cystic structures are observed in 14% of cases (115) and can be clearly visualized in sonography and MRI. Larger pseudocysts sometimes lend the tumor a blueish shimmer that can be seen during surgery. There are accounts of LG-ESS consisting almost entirely of pseudocysts (see below), though such cases are the exception (Fig. 4.1.9 (A), (C)D) (57, 215). Furthermore, tumors can also be very soft and largely decomposing. Different types of necrosis and focal hemorrhages are grossly discernible to varying degrees in all growth patterns, and often have equivalents in imaging diagnostic procedures (Fig. 4.1.10 (A)–(F), 4.1.12 (B)) (115).
Fig. 4.1.1: Low-grade endometrial stromal sarcoma – macroscopic findings; (A) nodulated tumor that is barely discernible from leiomyoma on gross observation; (B) large smooth formalin-fixated tumor that fills the entire uterine cavity; (C), (D) pedunculated lesion closely resembling leiomyoma that, like the tumor in (B), has prolapsed/is protruding from the cervix and exhibits expansive hemorrhages in its distal portions; (E) corresponding findings from vaginoscopy.
LG-ESS can also exhibit a less common growth pattern in which diffuse myometrial infiltration forces the myometrium apart/separates it. In such cases, the wall of the uterus appears enlarged and thickened, with a certain degree of nodulation. Directinfiltration of the ligaments and the tube are not uncommon in tumors exhibiting this growth pattern. VI can also be grossly discernible. There are multiple accounts of expansive intravascular tumors in the form of tumor thrombi (Fig. 4.1.3 (A), 4.1.15 (B), (C)) with growth into the vena cava (30, 193, 223). Such findings appear to be a consequence of the fact that the intravascular growth of LG-ESS into both lymphatic and blood vessels is cohesive, i.e. has direct communication to the main tumor (Fig. 4.1.3 (A), (B)). The difference to the discohesive intravascular growth exhibited by HG-ESS and UUS is clearly discernible (196) (see also Fig. 5.1.3 (D)). This cohesive intravascular growth is likely also the reason why LG-ESS only develop distant metastases rather late, and why distant metastasization is so rare in LG-ESS.
Fig. 4.1.2: Low-grade endometrial stromal sarcoma in macroscopy, microscopy and sonography; (A) cut-open LG-ESS specimen with what appears to be a capsule – in the left of the image – that gradually diminishes (or fades) towards the cut base and crosses over into infiltration on the opposite side that is clearly visible on gross observation; (B) corresponding histological aspect with extensive myometrial infiltration; (C) corresponding sonography of (A) with heterogeneous echogenicity and washy tumor margins.
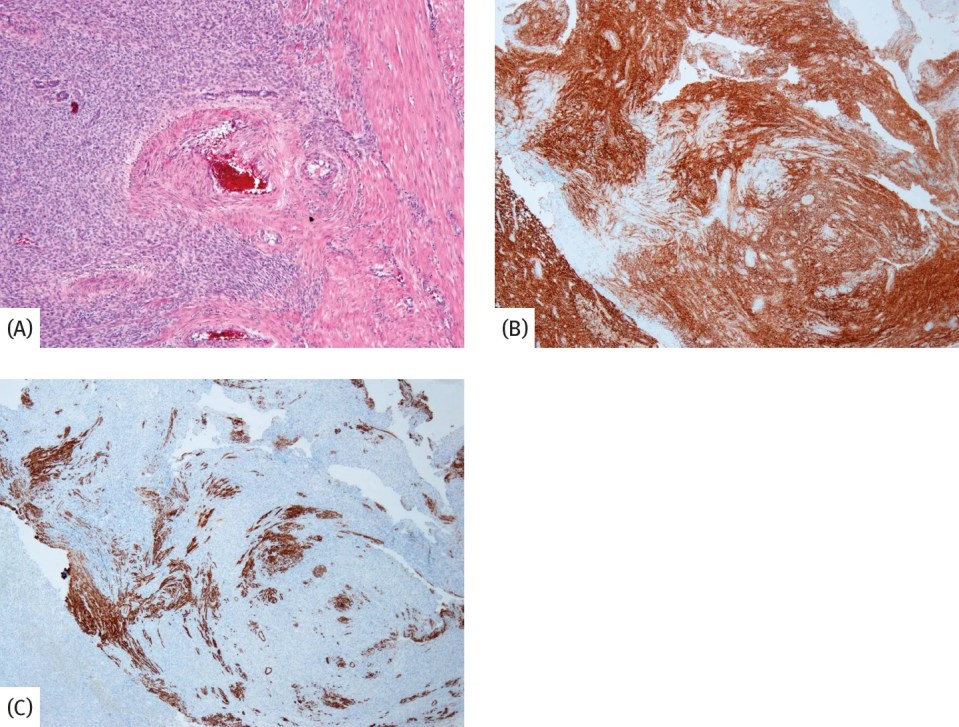
◂ Fig. 4.1.3: (A) worm-like intravascular tumor dissemination (tumor thrombus) of the hormone receptor positive low-grade component of a high-grade endometrial stromal sarcoma into the vena iliaca interna and communis. Corresponding features were also found at other localizations as well, (B) in low-grade endometrial stromal sarcomas, the intravascular invasion – into a lymphatic vessel in this instance – usually remains cohesively connected to the primary tumor and provides the foundation for extensive tumor thrombi on the basis of a tongue-like infiltration, as also depicted in Fig. 4.1.15 (A), (B). The endothelium of the lymphatic vessel is stained with the endothelial marker CD31. Tongue-like growth into the vessel at the narrow fissure between the infiltration and the actual tumor is recognizable at the center of the image (arrow). The wide connection to the stated tumor is clearly visible in the lower section of the image (arrow head).
LG-ESS with larger sections of myxoid differentiation usually have a gelatinous macroscopic appearance (168).
LG-ESS occasionally (albeit very rarely) also originate primarily in the cervix, though such tumors account for only 1.2% of all cervical sarcomas (22, 235). In contrast, 11.8% of corporal tumors exhibit cervical involvement (18).
Overall, LG-ESS consist of cells that are reminiscent of endometrial stroma in the proliferation phase. The cells cannot be morphologically discerned from ESN (cf. Chapter 3). Microscopically, LG-ESS are characterized by opulent small uniform cells with oval nuclei, small or absent nucleoli, scant cytoplasm, and poorly defined cytoplasm boundaries. Significant atypia are absent by definition (154). Accordingly, the microscopic appearance is monotonous and bland (Fig. 4.1.4 (A)).
Ample hyaline plaques are often visible and can serve as a helpful criterion for differentiation from smooth muscle tumors (241). LG-ESS can contain entrapped glands from the surrounding normal endometrium, especially at the margins of the tumor. Intra- and extrauterine LG-ESS with endometrial glands distributed either focally or across the entire tumor have been described in the literature (LG-ESS with extensive endometrioid glandular differentiation) (148). Such findings can serve to significantly exacerbate diagnostic differentiation from AS. Necroses and hemorrhages also occur in LG-ESS (231). LG-ESS can contain ample extensive pseudocysts that are usually lined with mesenchymal tumor cells (Fig. 4.1.9 (A), (D)).
Fig. 4.1.4: Low-grade endometrial stromal sarcoma – microscopic features. (A) the tumor usually consists of relatively monomorphic cells with scant cytoplasm that are usually arranged in an irregular, sometime fascicular fashion, and that can generally not be cytologically discerned from stromal nodules; (B) cytology is usually monotonous/bland, though mitoses (arrows) can sometimes be traced; (C) necroses (arrows) are rather less common; (D) in contrast, enclosures or “enclaves” of foam cells (vacuolized macrophages) constitute a common finding.
MI does not correlate with diagnosis and prognosis, and is usually measured at around < 5 M/10 HPF, but can also exceed 10 M/10 HPF. Atypical mitoses are rare, and true TCN hardly ever occur (Fig. 4.1.4 (B), (C)). The vessels most closely resemble uniformly distributed endometrial spiral arteries (Fig. 4.1.4 (A)). LVI and VI are observed in 19.1–26.6% (18, 115) resp. 30.8% of cases (115). The VI that LG-ESS exhibit differs from that of HG-ESS and UUS (72, 196). Compared to the aforementioned entities, LG-ESS harbor cohesive intravascular tumor foci with direct communication from the main tumor and attached to the vessel wall. The intravascular foci include tumor cells and small arteriole-type vessels and are surrounded by a thin fibrous band. Vascular markers confirmed the LVI and highlighted positively stained endothelial cells separating intravascular tumor foci from the blood itself. In contrast, intravascular tumor foci in HG-ESS are composed of discohesive cell clusters, lacking the features described in LG-ESS (Fig. 4.1.3 (A), (B)) (196). This feature can help to explain why LG-ESS metastasize much less commonly compared to HG-ESS. At the same time, it is also the cause of tumor thrombi in lymphatic and blood vessels that can reach considerable sizes (Fig. 4.1.3 (A), 4.1.15 (A), (B)) and that correspond to the worm-like invasion into the myometrium (72).
There is a strong likelihood that ESN constitutes a precursor for LG-ESS. Differentiating LG-ESS from ESN on the basis of specimens obtained via morcellation or curettage can be very difficult to nearly impossible if they contain no neighboring tissue. Often only focal, tongue-like permeative myometrial invasion (Fig. 4.1.5 (A)–(C)), LVI and also VI, stronger vascularity and more frequent endometrial involvement are the key differences between LG-ESS and ESN.
Fig. 4.1.5: Low-grade endometrial stromal sarcoma – differentiation from endometrial stromal nodules. (A) tongue-like myometrial invasion is the most important criterion for differentiation from stromal nodules. Said invasion can often not be verified in curettage or biopsy specimens, which is why final diagnosis can often only be reached on the basis of the hysterectomy specimen; (B), (C) myometrial invasions can occasionally already be immunohistochemically verified in specimens obtained from curettage; (B) the CD10 positive tumor cells overgrow CD10 negative leiomyocytes of the myometrium; (C) in this serial section of (B), the leiomyocytes strongly express caldesmon, while the tumor cells are caldesmon negative. (C) thus essentially constitutes the reverse or “negative” image of (B).
LG-ESS typically have a strong expression of ER and PGR (in 71–95% of cases) (20, 45, 94, 186), though PGR can be predominant (52, 94). It can so occur that only the PGRB isoform is expressed (20). ER and PGR are sometimes heterogeneously distributed (154). Absence of ER and PGR expression has been reported, albeit with ER being negativemore frequently than PGR (231, 237). AR are also positive in 45% of LG-ESS (152). LG-ESS can contain aromatases and can thus synthesize estrogens (187). GnRH receptor expression has also been verified in some cases. The HR provide the basis for therapy with progestins, AI and GnRH analogues. CD10 expression is strongly characteristic of LG-ESS, though not specific to them (Fig. 4.1.5 (B), 4.1.7(B)). Furthermore, in terms of myogenic markers, LG-ESS express vimentin, often express SMA and occasionally express desmin (44); h-caldesmon is not expressed. Up to 50% of LG-ESS also express cytokeratins (46). While one source states that LG-ESS do not express EGFR (176), another suggests that EGFR is expressed in up to 70% of cases (153). In another study, EGFR and PDGFR-β expression were immunohistochemically verified (96). VEGFR, PDGFR, c-Kit, p16, p53, and Ki67 are expressed in 26, 72, 82, 46, 10 and 54% of tumors (176). WT1 positivity is common (48), but Her-2/neu (10) is not overexpressed in LG-ESS. CD10 expression and the absence of h-caldesmon expression are diagnostically relevant features.
Like ESN, LG-ESS can also contain larger sections of myxoid and fibrous differentiation that can serve to spread the actual tumor cells far apart except for some focal accumulations. However, such tumors can usually nonetheless be classified as LG-ESS on the basis of their typical localization, their growth pattern, the presence of characteristic arterioles, the typical endometrial stroma and, not least, the lack of immunohistochemical verification of another type of differentiation (168). Rarer morphologic variants of LG-ESS include such in which oval to polygonal epithelioid cells with abundant eosinophilic cytoplasm account for 50 to 90% of the tumor cells (170).
Tumors with partial leiomyogenic differentiation constitute a much more important variant. Tumors are referred to as endometrial stromal sarcomas with mixed endometrial and smooth muscle differentiation when smooth muscle cells account for more than 30% of the tumor. The smooth muscle component is usually benign and typically expresses desmin and h-caldesmon. This notwithstanding, the tumor as a whole continues to have low malignant potential and its nature (in terms of benignity vs malignancy) does not differ from that of pure LG-ESS, i.e. said nature is likewise determined by the exhibited myometrial and vascular infiltration (154). Like ESN, LG- ESS can also contain sex cord-like elements. These tumors have been shown to express PGR B as the only or predominant progesterone isoform (52). In contrast, tumors with a myxoid endometrial stromal component, or with elements of smooth muscle differentiation and ample benign endometrioid-type glands that exhibit the typical features of IVLM, are extremely rare (147), as are LG-ESS with heterologous elements consisting of striated musculature, fat or bone (148).
Infiltration into surrounding tissues and structures constitutes the only microscopic difference between low-grade endometrial stromal sarcoma and endometrial stromal nodules. The morphologic aspect of the endometrial stroma is still microscopically discernible. Significant atypia are absent by definition and the mitotic index is usually lower than 10 M/10 HPF. CD10 constitutes the most important albeit non-specific immunohistochemical marker. Low-grade endometrial stromal sarcomas generally express estrogen and progesterone receptors.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree