Chapter 184 Kidney Stones (Renal Calculi, Nephrolithiasis)
 General Considerations
General Considerations
Stone formation in the urinary tract has been recognized for thousands of years; during the last few decades, however, both the pattern and incidence of the disease have changed markedly. In the past, stone formation occurred almost exclusively in the bladder, but today most stones form in the upper urinary tract. Males have a 3:1 ratio in the formation of kidney stones compared with females except in the sixth decade, where the incidence falls in men but rises in women—a trend toward gender equivalence.1
In the western hemisphere, over 80% of kidney stones are usually composed of calcium salts uric acid (5% to 8%), or struvite (10% to 15%). Molecular research is beginning to link certain mutations in genes responsible for handling renal chloride, which can lead to hypercalciuria. Other identified genetic changes are being linked to excess urinary excretions of oxalate, cystine, and uric acid.2 The incidence of renal stones varies geographically, reflecting differences in environmental factors, diet, and components of drinking water. Human urine is supersaturated with respect to calcium oxalate, uric acid, and phosphates. These substances normally remain in solution because of pH control and the secretion of inhibitors of crystal growth.
 Diagnostic Considerations
Diagnostic Considerations
Stone Formation
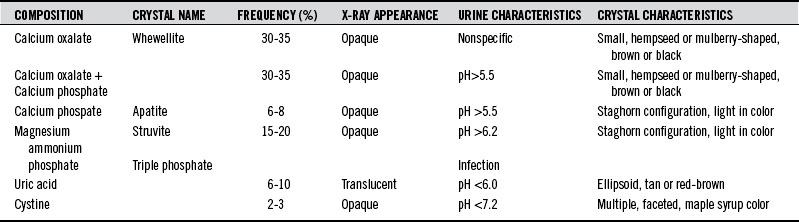
Conditions favoring stone formation can be divided into two groups: factors increasing the concentration of stone crystalloids and factors favoring stone formation at normal urinary concentrations. The first group includes reduction in urine volume (dehydration) and an increased rate of excretion of stone constituents. The second group is related to urinary stasis, pH changes, foreign bodies, and reduction of normal substances that solubilize stone constituents. See Tables 184-1 and 184-2 for an outline of the diagnostic possibilities.
TABLE 184-1 Causes of Excessive Excretion of Relatively Insoluble Urinary Constituents
| CONSTITUENT | CAUSE OF EXCESS EXCRETION | LABORATORY FINDINGS |
|---|---|---|
| Calcium | ||
| Oxalate | ||
| Uric acid | ||
| Cystine | Hereditary cystinuria |
TABLE 184-2 Physical Changes in Urine and the Kidneys
| CONDITION | POSSIBLE CAUSE |
|---|---|
| Increased concentration | |
| Urinary pH | |
| Infection | Proteus—struvite |
| Uricosuria | Crystals of uric acid initiate precipitation of calcium oxalate from solution |
| Nuclei for stone formation | |
| Deformities of kidney |
 Therapeutic Considerations
Therapeutic Considerations
Stone Composition
• Underlying metabolic or disease factors
• Serum and urinary calcium, uric acid, creatinine, and electrolyte levels
Table 184-3 summarizes the findings in the major types of kidney stones.
Dietary Factors
As a group, vegetarians have a lower risk of stone development. Studies have shown that even among meat eaters, those who ate higher amounts of fresh fruits and vegetables had a lower incidence of stones.10 Bran supplementation, as well as the simple change from white to whole wheat bread has resulted in lower urinary calcium levels.3
Dietary factors may also play a role in acidifying or alkalinizing urine. Depending on the type of stone, this ability to alter urinary pH may help to treat and prevent stones.11 In one study, 12 healthy men were given a standardized diet plus cranberry, black currant, or plum juice. These subjects then provided 24-hour urine collections for evaluation. The researchers found that cranberry juice decreased the urinary pH and significantly increased the excretion of oxalic acid and the relative supersaturation for uric acid. Black currant juice increased urinary pH, excretion of citric acid, and loss of oxalic acid. Plum juice effected no change.12 The researchers concluded that black currant juice could support the treatment and prevention of uric acid stones through its alkalizing effect. Conversely, because cranberry juice acidifies urine, it could be useful in the treatment of brushite and struvite stones as well as for urinary tract infections.
Increased fluid intake has been recognized as one of the main approaches to decrease urine supersaturation. An increase in the urine volume results in a decrease in stone prevalence. Numerous randomized controlled trials (RCTs) have found that consumption of more than 2 L/day of water13 or increased fluid intake to achieve a urine output of more than 2.5 L/day14 lowered the long-term risk of kidney stone recurrence by approximately 60% versus no treatment.
One trial, conducted in stone-forming men with a baseline soft drink consumption of more than 160 mL/day, reported a reduction in self-reported physician-confirmed episodes of renal colic in those randomized and advised to abstain from soft drink intake versus no intervention for 3 years. Total fluid intake was similar in both groups.9
 Diet
Diet
In one study, high levels of dietary calcium in men younger than age 60 were associated with decreased stone formation, but there was no effect in men older than age 60, nor was a supplemental dietary intake of more than 500 mg of calcium associated with an increased risk of stones.15
The specific type of stone was not elucidated in this study and it was assumed that the majority of kidney stones reported by the cohort population consisted predominantly of calcium oxalate, as in the general population. The cause of this age-specific difference is unclear. Dietary calcium may bind to dietary oxalate in the intestine, thereby reducing oxalate absorption and the subsequent concentration of urinary oxalate. Both vitamin D deficiency and a diminished ability to absorb dietary calcium are more prevalent in older people.15
Sodium and Salt
Regarding dietary salt content, observational studies suggested a strong relation between sodium consumption and hypercalciuria, but more recent evidence has challenged this idea. High sodium intake is known to reduce renal tubular reabsorption of calcium, thereby increasing the amount of calcium excreted in the urine.16 Also, a high-sodium diet is known to increase urine pH and has been proposed to reduce urine citrate.17 Other studies show a positive association between urinary sodium and calcium excretion and suggested that stone formers may be more sensitive to the calciuric effect of sodium.18
Nouvenne et al19 found that when patients with idiopathic calcium stones were treated with sodium restriction (60 mmol/day) and high fluid intake, a reduction of 100 mmol of urinary sodium was accompanied by a reduction of 64 mg/day in urinary calcium, with 30% of patients achieving normal urine calcium.
In contrast, other data, specifically in patients with hypocitraturia (not hypercalciuria), demonstrated that dietary sodium supplementation resulted in increased voided volume and decreased calcium oxalate supersaturation.20 This must be carefully examined, because these patients were followed for only a short period of time and they were already using pharmacologic interventions for stone disease. Although results continue to be conflicting, reduced dietary sodium is a general recommendation given to most patients with a history of kidney stones.
Fructose
Sports drinks have become increasingly popular within the past decade. The content of citrate in such beverages would be expected to increase urine citrate excretion and urine pH, providing protection against both calcium and uric acid stones. However, the purposely high sodium content of sports drinks, promoted as useful for “rehydration” in athletes, might be associated with increases in urine calcium excretion. In addition, the ingestion of significant amounts of sucrose and fructose in these drinks could also be associated with increases in calcium excretion.21 Fructose, a component of corn syrup frequently added to sports drinks and sodas as a sweetener, would also be an undesirable way for most stone formers to increase urine volume, given its recent links to hyperuricemia, metabolic syndrome, and stones.22,23
Citrate is an accepted inhibitor of the aggregation and growth of calcium oxalate and calcium phosphate crystals. In the past, dietary interventions to increase citrate have included lemonade and orange juice.24,25 The results with lemonade are conflicting, with one study showing an increase in urine volume but not in urine citrate.26 Regular consumption of grapefruit juice should be avoided, however. In one large-scale study, women drinking 8 oz of grapefruit juice daily increased their risk of stones by 44%.
Hyperoxaluria is a metabolic risk factor for stone disease. Dietary oxalate may contribute as much as 80% of the urine oxalate.27,28 Therefore, a low-oxalate diet may provide benefit for patients with hyperoxaluria. In addition, adequate dietary calcium to minimize intestinal oxalate absorption has been shown to be beneficial. Borghi et al29 in a randomized trial in men with recurrent calcium oxalate stones, achieved a significant reduction in oxalate excretion and the incidence of recurrent stones for patients on a normal-calcium (1200 mg/day), low-animal-protein, low-salt diet compared with a low-calcium diet (400 mg/day).
Dietary Recommendations for Patients with High Urine Oxalate
Low-Oxalate Diet
A low-oxalate diet is a common prescription for recurrent calcium oxalate kidney stones. The ultimate goal is to reduce the level of oxalic acid being excreted in the urine. It appears that people with recurrent kidney stones have a tendency to absorb higher levels of dietary oxalates compared with normal subjects not prone to kidney stones, who absorb only 3% to 8% of dietary oxalate. A low-oxalate diet is usually defined as less than 50 mg of oxalate per day, so foods in the high- and moderate-oxalate categories have to be curtailed. Box 184-1 is designed to provide an estimate of the oxalate content of food, which is highly variable. The level of oxalate in a particular food in published reports can vary twofold to fifteenfold. Differences in climate, soil quality, state of ripeness, or even which part of the plant is analyzed will also affect the value.