The biomechanical behavior of the intervertebral disk ultimately depends on the viability and activity of a small population of resident cells that make and maintain the disk’s extracellular matrix. Nutrients that support these cells are supplied by the blood vessels at the disks’ margins and diffuse through the matrix of the avascular disk to the cells. This article reviews pathways of nutrient supply to these cells; examines factors that may interrupt these pathways, and discusses consequences for disk cell survival, disk degeneration, and disk repair.
The normal adult human disk is virtually avascular. Nutrients, principally glucose and oxygen, necessary for survival and activity of disk cells are transported from blood vessels at its margins to the cells deep within the disk matrix, mainly by diffusion; products of metabolism, principally lactic acid, are removed by the reverse route. Loss of adequate nutrient supply has long been associated with the development and progression of disk degeneration because it adversely affects activity and even the viability of disk cells. Although the disk has only a small number of cells, these cells play a vital role. Such cells are responsible for making and maintaining the macromolecules of the disk matrix and, hence, ultimately govern the disk’s biomechanical functioning.
Here the authors discuss some of the factors that determine the rate at which nutrients can reach the disk cells. The authors review how these change with degeneration and age. The authors also discuss how the balance between rate of nutrient supply and rate of demand influences the nutrient-metabolite milieu of disk cells and, hence, their activity and survival. Finally, the authors assess the role of nutrient supply in developing successful biologic treatments for disk degeneration.
Factors influencing rate of supply of nutrients to the disk cells
Blood Supply to the Disks
The adult human disk is virtually avascular apart from a small number of capillaries that penetrate only a few millimeters into the outermost annulus. The disk is, thus, nourished by blood vessels at its margins. Those arising in the vertebral bodies feed most of the disk; only the outer annulus is nourished by blood vessels in the soft tissues at the annulus periphery. The supply through the vertebral bodies is, thus, critical for disk health. The blood supply to the vertebral column has been mapped at different stages of development and aging by several angiographic studies. In healthy young adults, the first through fourth lumbar vertebral bodies are supplied by pairs of arteries rising from the posterior wall of the abdominal aorta, whereas the fifth lumbar vertebral body is mainly supplied by arteries originating in the aortic bifurcation. These arteries branch into a complex system of smaller vessels that give rise to the capillaries responsible for supplying nutrients to the disk ( Fig. 1 ). These capillaries penetrate the marrow spaces of the subchondral plate of the vertebral body and terminate in loops at the interface between the vertebral body and cartilaginous end plate. They contain muscarinic receptors and, therefore, are not passive conduits 19 because flow through the capillary bed is regulated by drugs, such as acetyl choline and those in cigarette smoke, and possibly other physiologic signals, such as vibration. In addition to causing the capillary bed to constrict, these stimuli can also lead it to remodel in the long-term.
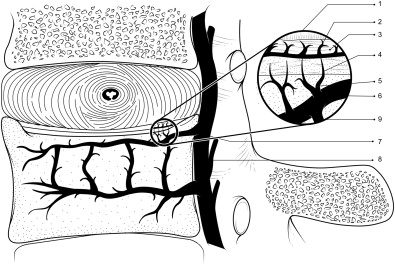
Transport of Nutrients from the Blood Vessels to the Disk Cells: Fluid Flow or Diffusion
The adult lumbar disks are large; the anterior height of the lower lumbar disks may be more than 20 mm and the mid-disk height more than 15 mm. Nutrients, thus, have to move long distances (about 5–8 mm) from the capillaries at the end plate-disk interface through a dense matrix to reach cells in the disk center.
Potentially, nutrients could move by diffusion under gradients set up by the cells’ metabolic demands. As is often suggested, nutrients could also be transported to the disk cells by fluid pumped in and out of the disk during customary daily changes in load. Although convective flow can enhance the transport of large molecules, several experimental and theoretical studies have shown that movement of small solutes, such as glucose and oxygen, through cartilage matrices is predominantly by diffusion rather than by convection. Indeed, if convection rather than diffusion played a major role in delivery of nutrients to the disk cells, the nutrient supply would be compromised during any prolonged period of inactivity; moreover, the cells would, in general, have a lower nutrient supply during the day’s activities when, as a result of the overall increased and sustained compression loading, there is a considerable net movement of fluid out of the disk.
The Cartilaginous End Plate
All nutrients supplied by the blood vessels arising in the vertebral body have to diffuse through the cartilaginous end plate, a thin layer of rigid hyaline cartilage lying between the vertebral body and the disk itself. In young humans, in addition to the layer of hyaline cartilage, there is a zone of growth cartilage adjacent to the bone, which is penetrated by cartilage canals facilitating nutrient transport into this area of the disk during growth. With the increase in maturity, the cartilage canals vanish and the growth cartilage thins to disappear in late adolescence. The disk then has to rely on nutrients supplied by small capillaries that penetrate the subchondral plate of the vertebral body and terminate in loops at the cartilage end plate (see Fig. 1 ) and possibly from transport via marrow spaces, which occupy around 20% of the cartilage/bone interface; these spaces seem directly related to permeability of the subchondral bone as measured in vitro.
Because all nutrients supplied by the capillaries of the end-plate route must pass through the cartilaginous end plate, transport properties of the end plate regulate movement of solutes into and out of the disk. As in other cartilages, penetration of solutes into the end plate depends on their charge, shape, and molecular weight; end-plate cartilage, thus, acts as a selective permeability barrier, allowing for easier passage of small solutes, such as glucose and oxygen, but severely impeding the transport of large solutes, such as growth factors and matrix macromolecules. Calcification of this end plate severely restricts the transport of even small solutes through the cartilaginous end plate as discussed later.
Diffusion Through the Disk Matrix
As in the cartilaginous end plate, movement of solutes through the disk itself is governed by factors that depend both on matrix composition and on solute properties. Solute movement through the matrix is restricted by its dense proteoglycan network, which regulates the size of pores available to solutes; the higher the proteoglycan concentration, the smaller the average pore size. In general, only a small fraction of pores are accessible to large solutes, limiting their concentration in the tissue to low levels. However, small solutes, such as oxygen, can penetrate into most of the available pores. As the disk degenerates and proteoglycans are lost, pore size increases, allowing access to large molecules, such as growth factors or cytokines, which are virtually excluded from normal disks.
The rate of movement of solutes through the matrix is regulated by solute diffusivity, which again depends on water content and matrix properties, particularly proteoglycan concentration. For instance, loss of hydration during loading, which increases proteoglycan concentration, will decrease diffusivity. Thus, for any solute, diffusivity varies with the position in the disk and depends on local proteoglycan concentration. In the annulus, diffusivity also depends on direction, differing radially from axially. Solute diffusivity also varies with solute molecular weight and shape. Diffusivities of small solutes, such as glucose and oxygen, are considerably greater than that of large proteins.
The amount diffusing into the disk is very sensitive to disk dimensions, particularly disk height ( Fig. 2 ). Diffusion to the center of the disk will be much more rapid in a small animal disk than in a large human lumbar disk ; and, thus, potential nutritional problems may be missed in animal disk degeneration models. In human disks, diffusion will also be affected by the load-induced variations in disk height because the disk may lose and regain more than 20% of its height during the diurnal cycle. Fluid loss increases proteoglycan content and, hence, decreases porosity, thus, leading to a decrease in solute diffusivity. It also, however, reduces disk height, potentially increasing the rate of transport to the disk center. Calculations and experiments show that these 2 opposing effects balance each other out for nutrients, such as oxygen and glucose, so that the diurnal loss and regain of fluid has little effect on nutrient concentration profiles, at least in normal disks. However, for solutes even as small as 500 daltons, long-term static loading seems to significantly impede transport into the center of the disks ; but it is unclear whether it is through alterations in blood flow or in transport through the matrix.
Failure of nutrient transport and association with disk degeneration
Disturbances of Nutrient Transport with Ageing and Pathology
Nutrients are supplied to the disk from capillaries, which are fed by blood flowing through the arteries of the vertebral bodies. Nutrients must then be able to pass from the capillaries through the cartilaginous endplate into the disk matrix. Several studies have shown that pathologic changes to any of these spinal structures can influence nutrient pathways and transport adversely and are associated with disk degeneration (see article by Chan and colleagues).
Although interest has focused on changes at the bone-disk junction, atherosclerosis of the lumbar arteries restricting blood flow to the lumbar spine has also been associated with disk degeneration and back pain. Kauppila has recently reviewed the evidence for this. For instance, chronic back pain was significantly more common in those with occluded or narrowed lumbar arteries ; calcification and stenotic changes seen in the abdominal aorta were associated with disk degeneration, as were high levels of cholesterol and triglycerides. However, it should be noted that disk degeneration is evident in many people in the first and second decade of life, well before atherosclerosis develops (see article by Chan and colleagues).
Disorders that affect the microcirculation, such as sickle-cell anemia, Gaucher disease, and Caisson disease, which all can restrict capillary flow, are also associated with disk degeneration. The effects of environmental factors, such as smoking, which can also restrict blood flow through the capillaries feeding the disk, are, however, controversial. Moreover, although several mechanisms linking smoking to disk degeneration have been proposed, there is no direct evidence to show that any association between smoking and disk degeneration is causal.
Pathologic changes that influence the pathway between blood supply and disk have long been shown to affect transport to the disk. Sclerosis of the bony end plate and increase in bone mineral density decrease contact area with the cartilaginous end plate and, hence, reduce end-plate permeability and are associated with an increase in disk degeneration. The cartilaginous end plate, a critical component in the transport pathway, shows early signs of degeneration with cracks, irregularities, protrusions of nucleus material through the end plate, and Schmorl nodes evident even by the second decade. Moreover, the end plate calcifies, which can severely impede transport from the blood supply to the disk. The degree of calcification of the end plate, as well as other irregularities, increases with ageing and also with disk degeneration ( Fig. 3 ). The cartilage end plates of scoliotic disks are also often heavily calcified, and here direct measurement has shown such calcification impedes transport from the blood supply into the disk.
Direct Visualization of Transport of Solutes into the Disk
Over the past decade, the development of relatively noninvasive magnetic resonance imaging (MRI) techniques for following movement of contrast medium has provided insights into how pathologic changes affect transport into the disk. In these studies, the enhancement of the signal in the disk following intravenous injection of a paramagnetic contrast agent is monitored over several hours ( Fig. 4 ). Comparison of the precontrast and postcontrast enhancement gives a semiquantitative measure of the rate of transport of the agent into the disk.
These studies have shown first of all that transport into disks is slow and in line with that expected from diffusion; the contrast agent moves forward as a diffusion front and only reaches the center of normal lumbar disks at around 6 hours. They have also shown definitively that degeneration of the disk affects transport of solutes into it; even with mild degeneration, transport is both reduced in extent and delayed. In degenerate disks, however, the pattern of enhancement is irregular and enhancement may be rapid with spikes within 10 minutes after injection, possibly because of vascular ingrowth and the breaches of the end plate. Rajasekaran and colleagues have devised an end-plate score, which relates to the extent of disk degeneration to patterns of postcontrast movement into the disk. This score could potentially be used as a diagnostic tool, but it needs further validation.
Because postcontrast serial MRI studies can be performed noninvasively and require only clinical MRI equipment, it is hoped that they increase our understanding of factors influencing nutrient transport and their relationship to disk failure. To date, postcontrast serial MRI has been used to study the effects of vasoactive drugs on nutrient transport. The administration of nimodepine, a vasodilator, increased signal intensity in the disk by approximately 15%, indicating the importance of blood flow rate on the delivery of nutrients to the disk. It has also been used to examine the effect of mechanical loading, whereby 4.5 hours of sustained creep loading reduced solute diffusion into the disk under static load. The reduction of transport into scoliotic disks, which undergo long-term sustained loading in vivo with consequent remodeling, was even more marked and was apparent before other MRI changes were visible.
The extracellular nutrient-metabolite milieu regulates cell viability and activity
As previously discussed, nutrients are transported to the disk cells from the arterial blood supply by a long and complex pathway that can be affected adversely by aging and degeneration. However, the local nutrient-metabolite milieu does not only depend on transport of nutrients to the cells but also by rates of cellular activity. It is this local milieu that determines cell function and, if nutrients or pH decrease to critical levels, cell survival. Cellular activity, particularly the production of matrix macromolecules, is strongly dependent on maintenance of an appropriate level of nutrients and metabolites. A decrease in oxygen tension, glucose levels, or in pH leads to a marked reduction in matrix gene expression and production of sulfated glycosaminoglycans, for instance. Furthermore, cells will not survive if glucose levels decrease less than approximately 0.5 mm or if lactic acid is not removed and consequently the extracellular pH decreases to less than approximately pH 6.7.
Rates of Cell Metabolism Regulate the Local Nutrient-Metabolite Milieu
The rates at which these disk cells consume nutrients, such as glucose and oxygen, and produce metabolic by-products, such as lactic acid, are critical determinants of the nutrient concentrations throughout the disk. For example, all disk cells consume sulfate to produce the sulfated glycosaminoglycan side chains of proteoglycans. However, actual rates of sulfate incorporation are very low compared with the concentration of sulfate available, so that sulfate concentration is hardly affected by cellular activity and the gradient is almost flat. For glucose, the situation is different, even though concentrations and diffusivities of sulfate and glucose are similar, disk cells consume glucose at rates that almost deplete it from the disk center. The rates of energy metabolism per cell vary with disk cell phenotype. Notochordal nucleus pulposus cells found in infant humans and in animals, such as rodents and pigs, consume oxygen and glucose at a much higher rate than the chondrocytelike nucleus cells seen in adult human disks. Moreover, other factors, such as the presence of cytokines and growth factors; mechanical stress; and levels of pH, oxygen, and glucose, also alter rates of nutrient consumption and lactic acid production. Thus, a disk that has an adequate nutrient supply under normal conditions, may be placed under nutrient stress by inflammatory signals or by inappropriate mechanical stresses.
Measured Nutrient-Metabolite Concentrations
There is little direct information on nutrient or metabolite concentrations in the disk because measurements of glucose, oxygen, lactate, and pH levels at the required sensitivity can now only be made invasively. Oxygen concentrations have been measured by inserting needle microelectrodes into disk explants in vitro and in vivo into disks of dogs ( Fig. 5 A) and human surgical patients. Intradiscal pH has also been measured in situ using electrodes. Lactate and glucose have, however, only been measured biochemically in explants or in tissue segments taken at surgery. All of these measurements confirm that there are concentration gradients of nutrients and metabolites in the disk, that these are consistent with those expected for transport of these solutes into and out of the disk by diffusion, and that pathologic changes can influence metabolite and nutrient concentrations considerably (see Fig. 5 B) and lead to situations whereby disk cells can no longer function or even survive.
The Importance of Modeling Nutrient Concentrations
It is difficult, if not impossible, particularly in humans, to obtain more than a limited snapshot of local levels of nutrients in the disk by direct measurement. Thus, measurement cannot provide an understanding of how ageing and pathologic changes or environmental factors, such as mechanical load, affect the extracellular nutrient-metabolite milieu. However, as previously discussed, nutrients are small molecules that move through the disk matrix almost entirely by diffusion under gradients set up by cellular metabolic activity. Levels of nutrients can, thus, be calculated using standard diffusion equations. The validity of diffusive models has been demonstrated in animal experiments in vivo and in model systems in vitro. In these cases, changes in concentration across the disk could be measured experimentally, and measured profiles were in good agreement with those calculated from diffusion-reaction models. Therefore, modeling is able to provide insight into nutrient-concentration profiles throughout the disk for situations whereby measurement is difficult, if not impossible.
The first models were analytical and, hence, were limited in scope. Nevertheless, they demonstrated how nutrient-concentration profiles were governed by the balance between rates of transport of nutrients into the disk and rates of cellular metabolism; thereby, cell density and the rate of metabolism per cell were critical parameters. A significant advance was provided by the incorporation of finite element methods, allowing calculation of profiles resulting from diffusion from both the endplate and the annulus periphery. A further advance in modeling arose when data on the interrelationships of metabolic rates became available. Metabolic profiles calculated using this information showed that profiles of nutrients and metabolites through the disk are interdependent ; hence, for example, in regions where oxygen tensions are low, glucose concentrations are necessarily also low and lactic acid concentrations high. Modeling has been further extended to examine diffusive profiles in a realistic 3-dimensional (3D) geometry. As such, the results demonstrate the importance of considering a 3D geometry for understanding details of the local nutrient-metabolite milieu, particularly in areas distant from the nutrient supply ( Fig. 6 ). A recent axisymmetric model that calculated effect of variations in end-plate permeability on nutrient and pH gradients used this information together with data on the critical nutrient concentrations necessary for cells to stay alive to calculate nutrient concentration levels, cell death, and viable cell densities in relation to end-plate properties.









