Chapter 77 Cimicifuga racemosa (Black Cohosh)
Cimicifuga racemosa (family: Ranunculaceae)
Common names: black cohosh, macrotys, rattleweed, black snake root
 Chemical Composition
Chemical Composition
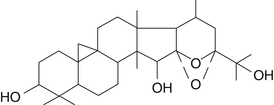
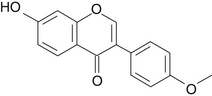
C. racemosa contains at least three important natural product groups that contribute to its pharmacology: cycloartane triterpene glycosides, such as actein and 26-deoxyactein; phenylpropanoid esters; and phenolic compounds, such as caffeic acid derivatives, including ferulic acid and isoferulic acid (Figures 77-1 and 77-2).1–3 Early reports showed that it also contained the phytoestrogen flavonoid formononetin, but more recent analysis demonstrated that this compound was not contained in either the crude herb or standardized extracts. At this point, despite extensive chemical, biological, and clinical studies, neither the active constituents nor the exact mode of action of black cohosh have been determined.4 Currently, the standardization of cimicifuga preparations is based on the content of triterpene glycosides, calculated as 26-deoxyactein. Other important triterpene glycosides include actein and cimicifugoside (aglycone cimegenol).
 Pharmacology
Pharmacology
The primary pharmacologic effects of cimicifuga appear to revolve around its ability to impact endocrine regulatory mechanisms.5–8 In the past, this activity was thought to be related to various phytoestrogenic components of cimicifuga, with formononetin perhaps being the most significant.9 However, this line of reasoning has been questioned by the lack of formononetin in clinically studied cimicifuga extracts, as well as equivocal reports on phytoestrogen activity. Current thinking is that certain cimicifuga components exert selective estrogen receptor modulator (SERM) activity with no action in the uterus, but that beneficial effects do occur in the hypothalamo/pituitary unit and in the bone.5,10,11
Cimicifuga’s primary effect on endocrine regulatory mechanisms appears to be the result of complex synergistic actions of its key ingredients. Evidence suggests that these compounds act on both the hypothalamus and vasomotor centers to produce significant clinical benefits in menopause. For example, one study to determine the endocrinologic actions of cimicifuga extract involved treating 110 women with either cimicifuga extract (supplying a total daily dose of 8 mg of 27-deoxyactein) or placebo.8 After 2 months of treatment, luteinizing hormone (LH) decreased by 20% in the cimicifuga group compared with the placebo group. Unlike estrogens, cimicifuga does not affect the release of prolactin and follicle-stimulating hormone. Researchers then divided the extract into three distinct types of active compounds on the basis of their ability to reduce LH secretion in ovariectomized rats and to compete in vitro with 17-β-estradiol for estrogen receptor binding sites, as follows:
• Constituents that did not bind to estrogen receptors but did suppress LH secretion
• Constituents able to bind to estrogen receptor sites and to inhibit LH secretion
• Compounds that did bind to estrogen receptors but did not inhibit LH secretion
Apparently, one of cimicifuga’s key pharmacologic effects is inhibition of the secretion of LH by the pituitary gland. This effect is accomplished equally by components that do and do not bind to estrogen receptors. If cimicifuga was simply mimicking the effects of estrogen, it would certainly alter the secretion of other pituitary hormones just like estrogen, but it does not. It appears that enhancement of dopaminergic activity is the responsible mechanism for some of these central effects on menopausal symptoms.5,12
Importantly, although early studies reported a direct estrogenic effect with cimicifuga, the current perspective is that ethanol and isopropanol extracts of black cohosh do not contain formononetin or other estrogenic flavonoids and, therefore, do not bind to the estrogen receptor, upregulate estrogen-dependent genes, or stimulate the growth of estrogen-dependent tumors in in vitro and animal models.5,13,14
 Clinical Applications
Clinical Applications
C. racemosa extracts are by far the most widely used and thoroughly studied natural alternatives to HRT in menopause. Clinical studies showed that cimicifuga extracts relieved not only hot flashes but also depression and vaginal atrophy.5,15–23
Although evidence indicates that cimicifuga may provide benefit in other gynecologic complaints such as premenstrual syndrome, amenorrhea (both primary and secondary), dysmenorrhea, polymenorrhea, uterine fibroids, and fibrocystic breast disease, its primary clinical application is menopause.22–26
Menopause
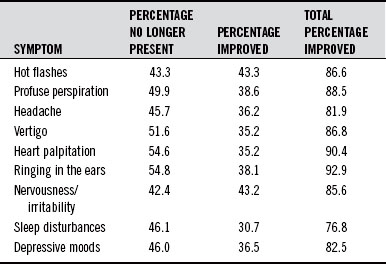
In one of the first clinical studies, a large open study involving 131 doctors and 629 female patients, cimicifuga extract (two tablets twice a day, providing a daily dose of 4 mg 27-deoxyactein) produced clear improvement of menopausal symptoms in more than 80% of patients within 6 to 8 weeks.18 As shown in Table 77-1, both physical and psychological symptoms improved.
In one of the first double-blind studies, 60 patients were given cimicifuga extract (two tablets twice a day providing a daily dose of 4 mg 27-deoxyactein), conjugated estrogens (0.625 mg/day), or diazepam (2 mg/day) for 12 weeks.19 Results from standard indexes of menopausal symptoms indicated a clear advantage of cimicifuga extract over both drugs. Cimicifuga’s effect on relieving depressive mood and anxiety associated with menopause was far superior to either diazepam or conjugated estrogens, as shown in Table 77-2.
TABLE 77-2 Effect on Kupperman Menopausal Index of Cimicifuga Compared with Conjugated Estrogens and Diazepam
| TREATMENT GROUP | BEGINNING | AT 12 WK |
|---|---|---|
| Cimicifuga | 35 | 14 |
| Conjugated estrogens | 35 | 16 |
| Diazepam | 35 | 20 |
One of the most used assessments in clinical studies in menopause is the Kupperman Menopausal Index. This quantitative assessment of menopausal symptoms is achieved by grading severity: severe = 3, moderate = 2, mild = 1, not present = 0. The symptoms assessed are the following:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree