Chapter 67 Artemisia annua (Sweet Wormwood)
Artemisia annua (family: Asteraceae)
Common names: sweet wormwood, sweet Annie, qing hao
 Chemical Composition
Chemical Composition
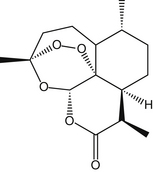
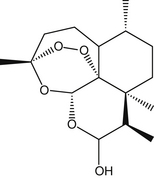
Sweet wormwood contains several sesquiterpene trioxane lactones, most notably artemisinin (also known as qinghaosu) but also deoxyartemisinin, artemisinic acid, and arteannuin-B.1 These compounds give the plant a distinctive bitter taste. A peroxide bridge in artemisinin is vital in the antimalarial effects of the compound (Figure 67-1).2 Artemisinin is rapidly and readily reduced to dihydroartemisinin in the body. Dihydroartemisinin appears to be the active metabolite of artemisinin and all synthetic artemisinin derivatives developed to date (Figure 67-2).3
Sweet wormwood also contains a number of flavonoids.1 In vitro research suggests these flavonoids potentiate the antimalarial activity of the sesquiterpene lactones.4
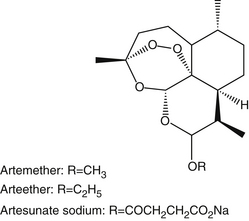
Three semisynthetic derivatives of artemisinin are widely available for use: artemether, artesunate, and arteether.3 Artemether is a lipophilic β-epimer of dihydroartemisinin and is administered intramuscularly. Artesunate is a hydrophilic hemisuccinate compound administered orally or by injection. Arteether is an ethyl ether derivative of dihydroartemisinin and is lipophilic (Figure 67-3).
 History and Folk Use
History and Folk Use
The herb, known to the ancient Chinese as qing hao, was recommended for treatment of fever as early as 341 AD, as written in the handbook for prescription for emergencies, Zhou Hou Bei Ji Fang by Ge Heng.3 Qing hao had been prescribed in traditional Chinese medicine for other problems for at least 500 years beforehand on the basis of extant written records.
Qinghaosu, known as artemisinin in the West, was isolated and identified in qing hao in the 1970s in China.5 It was thoroughly investigated and determined to have excellent antimalarial activity.
 Pharmacology
Pharmacology
Antimalarial Effects
Dihydroartemisinin preferentially accumulates in erythrocytes, and malaria-infected erythrocytes accumulate 300 times as much as uninfected erythrocytes.6 Because active artemisinin compounds all appear to be converted to dihydroartemisinin in vitro and then act as mentioned previously, all these compounds ultimately end up in erythrocytes.
Artemisinin and related molecules appear to act in part by generation of free radicals, leading to damaging the membranes of several parasitic organisms.7 This leads to rapid death of all stages of malaria parasites.8 Because the presence of an oxidizing peroxide bridge appears essential to the activity of artemisinin compounds, the oxidative hypothesis is further supported. The importance of iron to this process is unclear. Some investigators suggest that iron chelators such as desferrioxamine actually increase the activity of artemisinin in vitro.9
Artemisinin compounds are synergistic with many other, but not all, antimalarial drugs. Low doses of mefloquine and artemisinin showed definite synergism in vitro against Plasmodium falciparum, whereas higher doses showed additive effects.10 Mefloquine, chloroquine, primaquine, and tetracycline were all potentiated by artemisinin in mice with malaria; the effects of pyrimethamine, cycloguanil, and sulphonamide antibiotics were all decreased.11
Antiparasitic Effects
Besides killing Plasmodium spp., artemisinin compounds have shown activity against Schistosoma spp. and Clonorchis sinensis. Numerous animal studies show that artemether and artesunate can prevent and cure infections by Schistosoma japonicum, Schistosoma mansoni, and Schistosoma haematobium.12–15 It is not clear if artemisinin or dihydroartemisinin is effective. However, artemisinin and the various available semisynthetic variants were all effective at treating Clonorchis infections in rats with no sign of toxicity.16
Antineoplastic Effects
Mounting evidence suggests that artemisinin compounds have potent anticancer activity. Artemisinin and quercetagetin 6,7,3’,4’-tetramethyl ether, a flavonoid found in sweet wormwood, both showed activity against several tumor types in vitro.17 Dihydroartemisinin and artesunate have also been investigated and found to inhibit cancer cells significantly in vitro.18,19 The presence of ferrous iron appears to be essential for this activity, and sufficient quantities of iron may be necessary to prevent cancer cells from becoming resistant to artemisinin compounds.18,20
A combination of ferrous sulfate and dihydroartemisinin proved effective at limiting the growth of implanted fibrosarcomas in rats.21 The combination did not appear to cause adverse effects. Ferrous sulfate or dihydroartemisinin administered alone were not effective in this model.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree