Treatment of Congenital Femoral Deficiency
Dror Paley
DEFINITION
The term proximal focal femoral deficiency (PFFD) is used to describe congenital femoral deficiency and deformity of the proximal femur to be distinguished from the congenital short femur.6 However, the more comprehensive term congenital femoral deficiency (CFD)15 better describes the spectrum of deficiency, deformity, and discrepancy ranging from the congenital short femur to the most severe PFFD.
The severity of the deformity varies widely, and this condition can be diagnosed in the prenatal period using ultrasound examination.12
In most cases, CFD is not simple coxa vara. Patients with CFD lack integrity, stability, and mobility of the hip and knee, with concurrent joint malorientation, bony deformity, and soft tissue contractures. The affected limb grows at an inhibited rate depending on the severity of the underlying deficiency. The resulting limb length discrepancy (LLD) can be accurately predicted using the multiplier method.1, 2, 11
ANATOMY
Although existing classification systems for PFFD are descriptive, these classification systems are not helpful in determining the final femoral morphology or treatment strategies.15
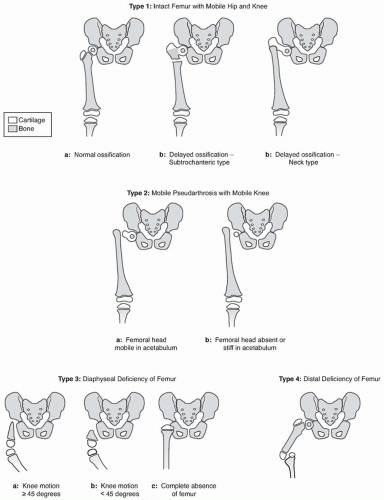
The Paley classification system (FIG 1) is based on factors that reflect the severity of pathology and reconstructability of the congenitally deficient femur. This classification is based on pathologic factors that determine surgical reconstruction strategies.9
The abnormal anatomy of CFD consists of coxa vara of the proximal femur with abduction contracture of the hip (ie, tensor fascia lata [TFL], gluteus maximus, gluteus medius, and gluteus minimus muscles), proximal femoral flexion deformity with concurrent hip flexion contracture (ie, rectus femoris, TFL, iliopsoas, gluteus medius and minimus muscles), and external femoral torsion-retroversion with concurrent external soft tissue contracture (ie, piriformis muscle).
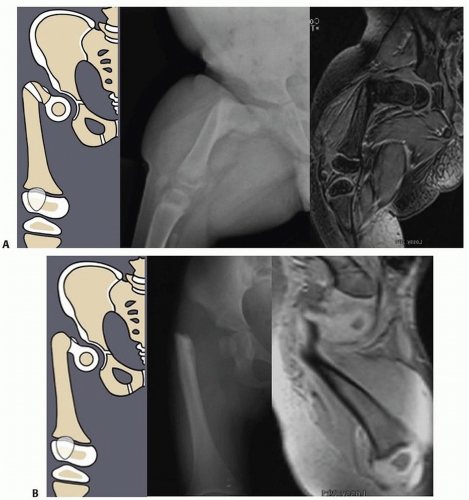
The best way to understand the proximal femoral deformity of the more severe cases is to imagine creating the deformity de novo: start with a proximal femoral segment that ends in the subtrochanteric region. Perform all of these movements relative to the coronal plane of the pelvis in its normal anatomic position:
First, flex the proximal femur 90 degrees.
In the flexed position, abduct the proximal femur 45 degrees.
Connect the distal femoral diaphyseal segment to it with the distal femur rotated externally 45 degrees relative to the pelvis.
The resulting deformity is that seen with severe CFD (FIG 2).
The proximal femur can also present a region of delayed ossification in either the subtrochanteric region or the neck region or both. Ossification of the cartilaginous proximal femur differentiates Paley type 1a CFD (ie, normal ossification) from Paley type 1b CFD (ie, delayed ossification). The latter can be subclassified as Paley type 1b subtrochanteric type, neck type, or combined subtrochanteric-neck type (see FIG 1).
Once treated with realignment and in some cases insertion of bone morphogenetic protein (BMP), the unossified cartilage of the proximal femur in the type 1b will ossify changing the femur into type 1a. This area of delayed ossification is often mistaken for a pseudarthrosis (it could be referred to as a stiff cartilaginous pseudarthrosis to differentiate it from type 2, in which there is a true mobile, fibrous pseudarthrosis).
A more severe form of CFD is classified as Paley type 2; this type has a true mobile pseudarthrosis between the greater trochanter and femoral head or complete absence of the femoral head (see FIG 1).
The most severe proximal deficiencies are classified as Paley type 3 (diaphyseal deficiencies). In these cases, the greater trochanter is absent and the knee joint is affected to a greater (range of motion [ROM] <45 degrees) or lesser (ROM more than 45 degrees) extent. Complete absence of the femur is included in this group.
In very rare cases, there is a distal deficiency of the femur (ie, Paley type 4). Cases of distal deficiency present with very severe knee varus but a well-developed, intact hip joint.
Acetabular dysplasia is almost always present in patients with CFD. This deformity must be recognized and corrected to prevent subluxation or dislocation of the hip during lengthening.
Congenital knee abnormalities also exist with CFD. Absent or hypoplastic cruciate ligaments (ie, anterior cruciate ligament [ACL], posterior cruciate ligament [PCL]); hypoplastic lateral femoral condyle resulting in genu valgum; and hypoplastic patella with lateral maltracking, subluxation, or dislocation are common. Rotatory instability of the tibiofemoral joint and knee flexion contractures (ie, biceps femoris muscle, posterior knee joint capsule, iliotibial band) are also common.
PATHOGENESIS
The cause of an isolated single-limb abnormality remains unknown in most cases. Unilateral CFD is usually not related to a genetic syndrome. Bilateral CFD, multiple limb deficiencies, and associated tibial hemimelia are usually related to a genetic origin.
A patient with CFD presenting for initial evaluation does not require a genetic consultation unless multiple limb deficiencies or other congenital malformations are present.
NATURAL HISTORY
The natural history of CFD is a progressive LLD in unilateral cases. The deformities and soft tissue contractures described above persist but do not progress.
The Paley type 1b hip shows eventual ossification of the cartilaginous femoral neck or subtrochanteric region.15 Although ossification occurs over time, the bony deformities and soft tissue contractures persist.
Progressive LLD can be accurately predicted using the Paley multiplier method.1, 2, 11 Determining the LLD at maturity and using the Paley classification system allows the surgeon to formulate an overall strategy for deformity correction and limb lengthening.
The number and timing of surgical procedures can be presented as a general overall plan to the parents during the initial consultation.
PATIENT HISTORY AND PHYSICAL FINDINGS
A general history and physical examination should be performed.
The clinician should concentrate on family history or concurrent known congenital abnormalities, which could indicate a genetic syndrome that could require further workup and genetic consultation.
The facies, upper extremities, and spine are also examined, looking for abnormal appearance or multiple congenital anomalies, which can indicate a genetic syndrome. In such cases, genetic consultation should be obtained.
Hip ROM
Abduction-adduction and flexion ROM are examined in the supine position. Thomas test (hip extension) is performed to measure fixed flexion deformity of the hip. Hip internal rotation-external rotation is measured in the prone position, together with the thigh-foot angle to determine a rotation profile for the femur and tibia, respectively. Muscle length tests include popliteal angle (hamstring length), prone knee bend Ely test (rectus femoris muscle), and Ober test (tensor fascia lata-iliotibial band). The latter is performed with the patient in the lateral decubitus position pulling the hip into extension with the knee flexed to 90 degrees. If the leg does not drop into adduction by gravity, there is a positive Ober sign, indicating a contracture of the TFL and iliotibial band.
ROM is measured, and contractures are identified and quantified in degrees. A popliteal angle of more than 0 degree
and prone knee bend less than supine knee bend indicate tightness of the hamstring and rectus femoris muscles, respectively. A positive Ober sign is almost always present.
Contractures need to be treated in preparation for lengthening. Lengthening of the rectus femoris and hamstring muscles is recommended for positive muscle tightness. Lengthening or excision of the fascia lata and iliotibial band is always recommended prior to or at the time of femoral lengthening.
Knee ROM
Flexion and extension knee ROM is measured in the supine and prone positions.
Greater than 10 degrees of fixed flexion deformity should be corrected during preparatory procedures. A fixed flexion deformity may be present.
Knee stability (anteroposterior [AP])
The Lachman test and the anterior and posterior drawer tests are performed. The clinician looks for posterior sag and rotatory instability. The amount of instability is measured:
Grade I: mild with end point
Grade II: moderate with end point
Grade III: moderate or severe with no end point
AP knee instability is common. It is often difficult to tell if the instability is anterior, posterior, or both.
Knee stability (rotatory)
The rotatory stability of the knee joint is examined by internally and externally rotating the tibia on the distal femur in flexion and extension. The presence of subluxation with rotation of the tibia on the distal femur is noted.
External rotatory instability is a common finding that is secondary to a contracted iliotibial band and biceps femoris tendons and can lead to rotatory subluxation of the knee and patellar dislocation.
Patellar stability
The clinician should flex the knee and palpate the alignment of the patella to the notch in flexion. Tracking of the patella is assessed from 0 to 90 degrees. The clinicians should attempt to push a thumb into the intercondylar notch.
If the examiner’s thumb is able to palpate the intercondylar notch with the patient’s knee flexed, this denotes lateral subluxation or dislocation of the patella.
Patellar instability is common and can be an indication of lateral rotatory instability of the knee and contracture of the iliotibial band.
The clinician should look at the overall appearance of the foot and ankle.
Any missing rays or positional abnormalities are noted. Ankle ROM is tested with knee flexed and extended. Inversion and eversion ROM is tested.
The amount of dorsiflexion, plantarflexion, inversion, and eversion is recorded. Equinovalgus deformity with missing lateral rays indicates concurrent fibular hemimelia. Subtle increase in eversion ROM indicates fibular hypoplasia or a ball-and-socket ankle joint.
IMAGING AND OTHER DIAGNOSTIC STUDIES
During the initial evaluation of an infant with CFD, pull down supine long AP and lateral view radiographs should be obtained that include the pelvis and both lower extremities. Both lower limbs are “pulled down” to make sure both knees are in maximum extension (FIG 3).
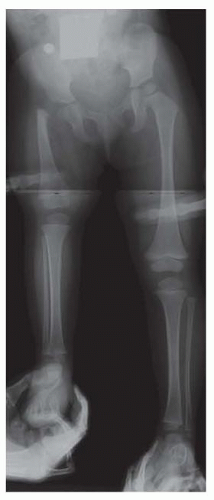
FIG 3 • Pull down view long x-ray to assess leg length difference in infancy. Note the hands of the parent.
The supine, long AP view radiograph should be assessed for the overall appearance of the ossific anatomy. This radiograph should allow the physician to classify the type of CFD.
The lengths of both femora and both tibiae should be measured. The difference between them is the LLD, not including the foot. The clinicians should measure from the lateral acetabular edge to the midpoint of the knee joint space for the femoral lengths and from the same midpoint of the knee joint space to the end of the talar ossific nucleus for the tibial lengths. The amount of current LLD can be used with the multiplier method to predict the overall LLD at maturity.1, 2, 11
The acetabulum should be assessed for dysplasia using the center-edge (CE) angle (even in infants) and the acetabular index (AI).
The long lateral view radiograph of the lower extremity is assessed for underlying fixed flexion deformity of the knee.
The anterior cortical line of the distal femur should normally be colinear with the anterior cortical line of the proximal tibia. A flexion angle between these lines represents fixed flexion deformity of the knee. It is important that the lateral x-ray be taken with the patella forward to avoid mistaking knee valgus for flexion (the valgus of an externally rotated knee will appear to be flexed).
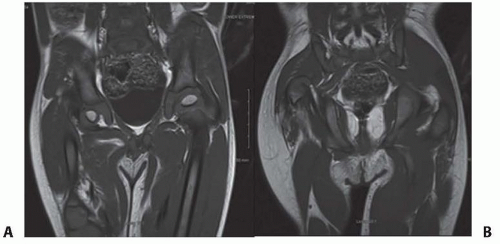
Other imaging studies that are useful include magnetic resonance imaging (MRI) and arthrography of the hips. All Paley types 1b and 2 should have an MRI after age 18 months to confirm whether there is a cartilaginous connection between the femoral head and shaft (FIG 4).
Arthrography under general anesthesia is also helpful to determine the presence of pseudarthrosis versus delayed ossification of the proximal femur. Although the arthrogram is obtained, the lower extremity is manipulated and the proximal femur is visualized.
If the proximal femur and femoral head move as a unit, this usually denotes a cartilaginous connection
in the proximal femur, and the CFD is classified as type 1b. The arthrogram is also useful to differentiate between Paley types 2a and 2b. Both 2a and 2b might have a femoral head present; the difference is whether the femoral head is fused to the acetabulum or not. If dye can be injected into a joint space, the hip can usually be classified as type 2a. In some cases of type 2a, the dye can be seen to outline a cleft through the femoral neck.
DIFFERENTIAL DIAGNOSIS
If the patient has bilateral CFD, the clinician must consider the following differential diagnoses:
Camptomelic syndrome
Femoral hypoplasia with unusual facies syndrome
NONOPERATIVE MANAGEMENT
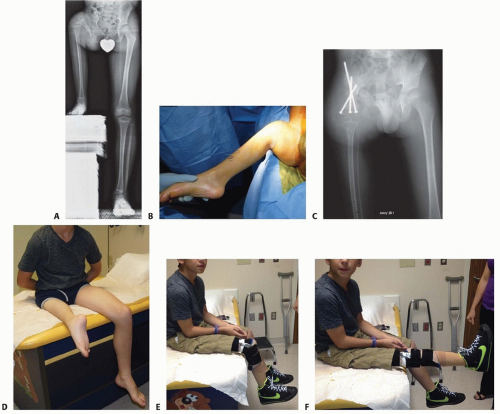
Shoe lifts, orthoses, and prostheses are used for the nonoperative management of LLD. All children should receive a shoe or prosthesis with a lift when they begin to cruise the furniture. A simple shoe lift of an amount equal to 1 cm less than the LLD is used in most cases in which LLD is less than 10 cm (FIG 5).
It is helpful to supplement the lift with an articulated ankle-foot orthosis (AFO) for ankle support from the long lever arm of the shoe lift. If the lift is more than 10 cm, a prosthetic foot connected to an articulated AFO is preferred both to reduce weight and improve cosmesis.
The clinician should avoid splinting the foot in equinus because it might cause an equinus contracture.
In children younger than 6 years, a limb length radiograph should be obtained every 6 months to assess LLD and prescribe a new lift.
After age 6 years, annual assessment and prescription is adequate.
In more severe cases with hip and knee fixed flexion deformity, it might be necessary to extend the orthotic or prosthetic support above the knee with ischial bearing support.
SURGICAL MANAGEMENT
Patients with types 1a, 1b, 2a, and 2b CFD can be managed successfully with lengthening reconstruction surgery as opposed to prosthetic reconstruction surgery.
Before undergoing lengthening reconstruction surgery, patients with certain knee and hip deformities and deficiencies should undergo preparatory procedures to prevent complications during lengthening and to reconstruct the knee and hip joints. This chapter will present the preparatory surgical procedures of the hip and knee and the external fixation method we prefer for CFD lengthening surgery.
Type 1 Congenital Femoral Deficiency
Type 1 CFD is the most reconstructable.
Before lengthening, hip stability should be determined radiographically. The best indicator is the CE angle. If the CE angle is less than 20 degrees, a Dega osteotomy should be performed before lengthening. In addition, the AI should be less than 30 degrees. If the CE angle is borderline 20 degrees but the AI or inclination of the sourcil is high, it is better to err on the side of caution and perform a Dega osteotomy (FIG 6).
Coxa vara should be corrected before lengthening if the neck-shaft angle is less than 120 degrees. When coxa vara and hip dysplasia are present and when the coxa vara is severe, the superhip procedure is performed. The pelvic and femoral osteotomies should be performed 12 months before the first lengthening. The superhip procedure is a
comprehensive surgery to correct the proximal femoral and hip deformities with concurrent soft tissue releases.
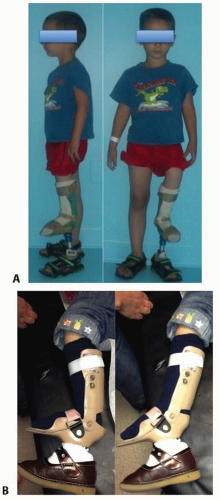
FIG 5 • A. Articulated AFO prosthesis. B. Plantar flexion and dorsiflexion. The ankle motion aids with walking.
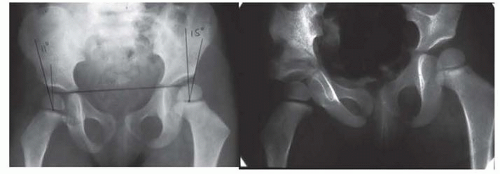
FIG 6 • Paley type 1a CFD with dysplastic acetabulum. The CE angle is 11 degrees. The patient underwent a Dega osteotomy.
At the conclusion of a successful superhip procedure, the proximal femur has been anatomically and biomechanically reconstructed and the delayed ossification of the femoral neck has ossified. The femur can now be reclassified from 1b to 1a. This ossification occurs within 3 to 12 months of the superhip procedure (often aided by the insertion of BMP). Lengthening is usually not performed in type 1b cases until they convert to type 1a. If lack of full ossification of the femoral neck or subtrochanteric region persists, despite the superhip procedure, repeat insertion of BMP is indicated. As a final measure to allow lengthening to begin in such hips at risk, the external fixation can be extended to the pelvis to protect the hip.
Types 2 and 3 Congenital Femoral Deficiency
The strategies that should be used to treat types 2 and 3 CFD are complex and beyond the scope of this chapter. A summary of the strategies is provided in the following text.
Type 2 Congenital Femoral Deficiency
The presence or absence of a mobile femoral head in the acetabulum determines the treatment strategy. MRI will show if the femoral head is fused to the acetabulum by a bone or cartilage bridge. If a fusion is present, it is always between the femoral head and the posterior wall of the acetabulum (ischium).
If the femoral head is mobile, it can be connected to the remainder of the femur by a complicated procedure in which the femoral neck is reconstructed (superhip 2 procedure). If the femoral head does not move in the acetabulum, there are three options:
Separate it from the acetabulum creating a mobile head and proceed with a superhip 2 procedure.
Enucleate the femoral head from the acetabulum and perform a superhip 3 procedure.
Do a soft tissue release and lengthen the femur. Near skeletal maturity perform a pelvic support osteotomy.13
The superhip 2 procedure converts types 2a and 2b CFD to type 1a. The proximal femur including the greater trochanter is converted into a femoral neck. An osteotomy is performed just distal to the psoas tendon insertion and the bone rotated 135 degrees. All the muscles except the quadriceps are detached from the proximal femur. The quadriceps acts as the vascular pedicle for this segment of bone. The distal diaphysis is osteotomized at a 45-degree angle and shortened to allow fixation to the new neck segment. This produces a femur with a femoral neck-shaft angle of 135 degrees. The new neck is connected to the ossific nucleus of the femoral head with threaded k-wires or a screw. All of the muscles are reattached to the femur.
If the femoral head is not worthy of reconstruction, a superhip 3 procedure can be performed instead. This is a form of trochanteric arthroplasty. The quadriceps again serves as the vascular pedicle. All other muscles are detached. The femur is osteotomized and shortened. The hooked greater trochanter fits nicely in the acetabulum. If the acetabulum needs to be enlarged to accommodate the trochanter, the bone distal to the triradiate cartilage is burred allowing the triradiate to act as part of the dome. The anterior ischium is also burred to deepen the posterior wall, and interposition capsule is applied over the exposed bone.
Type 3 Congenital Femoral Deficiency
Type 3a can be treated like type 2b. Patients can undergo hip release, serial lengthenings, and pelvic support osteotomy or superhip 2 or 3 followed by serial lengthenings or they can be treated by prosthetic fitting options, including prosthetic reconstruction surgery (ie, Syme amputation or rotationplasty3).
Prosthetic reconstruction surgery is recommended for most type 3 CFD due to the extensive deficiency present. This is especially the case for type 3b because there is a stiff knee joint (<45 degrees of motion). Although type 3a can be converted to type 2b, the treatment would consist of four or more lengthenings. Rotationplasty is recommended for most type 3a because it provides a more predictable functional result than does lengthening (FIG 7).
Lengthening
The number of lengthenings that are required for type 1 CFD is determined by the initial LLD prediction. Patients that do not require any preparatory surgery can undergo their first lengthening as early as age 2 years and preferably before age 4 years. Patients that do require preparatory surgery (eg, superhip, Dega) can undergo their first lengthening 1 year or more after the preparatory surgery. This is usually between ages 3 and 4 years. Between 5 and 8 cm can be obtained during each lengthening.
For type 1 CFD, the femur should be lengthened by using a distal femoral osteotomy instead of a proximal femoral osteotomy.
Distal osteotomies allow for better regenerate bone formation because they have a broader cross-sectional diameter and because the bone is not sclerotic or dysvascular, which often is the case in the proximal femur of patients with CFD. Distal osteotomies can also be used to simultaneously correct the valgus deformity of the distal femur.
Proximal osteotomies are used to correct the external femoral torsion and proximal varus deformities and are usually part of the preparatory surgery and not the lengthening surgery. Proximal osteotomies are not used for lengthening because of poor regenerate bone formation. A proximal osteotomy can be used for deformity correction with a concurrent distal osteotomy for lengthening.
Lengthening via External Fixators
Femoral lengthening with an external fixator can be performed with various devices.
The essential principle of lengthening with external fixation is to stabilize the knee during lengthening while allowing for knee motion. This is accomplished by using hinges and external fixation of the tibia.
From 1987 to 2000, the author used only the Ilizarov apparatus with fixation across the knee joint with a hinge for all CFD lengthening cases. This method has previously been described.9 A monolateral external fixator was not used because it could not articulate across the knee joint.
From 2000 to 2009, the author modified the Orthofix Limb Reconstruction System (LRS) rail (Orthofix, Inc., McKinney, TX) with the Sheffield Ring Fixation System arch (Orthofix) to articulate across the knee with fixation to the tibia. This method was used for all CFD cases between January 2000 and May 2009.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree