Sleep patterns share common pathways with nociceptive stimuli. Several important factors are reviewed in considering connections between sleep and pain. Causes for sleep fragmentation include sleep disordered breathing; abnormal leg movements, including restless legs syndrome and periodic limb movements; and underlying mood disorder, which may be exacerbated by physical symptoms. Identification and management of insomnia includes the definition of the condition, pharmacologic interventions, the role of circadian rhythms and clock adjustments, and the use of cognitive behavior therapy for insomnia.
Key points
- •
Sleep patterns share common pathways with nociceptive stimuli.
- •
Causes for sleep fragmentation include (1) sleep disordered breathing; (2) abnormal leg movements, including restless legs syndrome, occurring while the patient is awake, and periodic limb movements that occur while the patient is asleep; (3) underlying mood disorder, which may be exacerbated by physical symptoms; (4) hormonal changes.
Summary and objectives
- 1.
Sleep patterns share common pathways with nociceptive stimuli. Several important factors are reviewed in considering connections between sleep and pain.
- 2.
Causes for sleep fragmentation include:
- a.
Sleep disordered breathing, which may present with snoring, witnessed apneas, daytime sleepiness, and also with more subtle symptoms like morning headache and anxiety. Home sleep testing or more extensive in-laboratory polysomnography may be used for diagnosis of this condition. Treatment options include use of continuous positive airway pressure (CPAP); oral advancement devices (OAD); weight loss; surgical interventions; and, most recently, US Food and Drug Administration (FDA)–approved upper airway stimulation devices.
- b.
Abnormal leg movements, including restless legs syndrome (RLS), occurring while the patient is awake and periodic limb movements that occur while the patient is asleep.
- c.
Underlying mood disorder, which may be exacerbated by physical symptoms.
- a.
- 3.
Identification and management of insomnia includes the definition of the condition, pharmacologic interventions, the role of circadian rhythms and clock adjustments, and the use of cognitive behavior therapy (CBT) for insomnia.
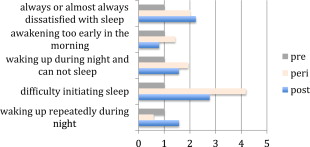
Pain and sleep disorders share a reciprocal relationship: pain interferes with sleep quality and, in turn, poor and truncated sleep perpetuates pain symptoms. Furthermore, nociceptive pathways and sleep-wake pathways may share common central serotonergic transmission. A survey of 18,980 individuals from 5 European countries showed that significantly more participants with chronic painful conditions (eg, limb or joint pain, backache, gastrointestinal pain, and headache) than those without pain experienced insomnia. Compared with individuals without chronic pain conditions, those with pain were 3 times more likely to report difficulties with initiating sleep, maintaining sleep, early morning awakenings, and nonrestorative sleep. There may be confounding factors, such as underlying mental illness and the onset of menopause. These confounding factors are often important and need to be considered when addressing sleep difficulties in patients with pain. Menopause is an important physiologic change in women that has both physiologic and psychological implications. As Fig. 1 shows, sleep difficulties arise during this period that may layer on the nocturnal symptoms associated with pain.
Average adults require 7 to 8 hours sleep, with less than 6 and more than 9 hours correlating with adverse health outcomes. Insomnia encompasses the inability to initiate sleep, maintain sleep, or reach a state of restfulness and refreshment on awakening. It may be associated with daytime symptoms of fatigue, memory deficits, social/vocational/academic performance deficits, mood changes, daytime sleepiness, lack of motivation, vulnerability to accidents, somatic symptoms, and a preoccupation with sleep that perpetuates the cycle of insomnia. Thirty percent of the working population in the United States sleeps for less than 6 hours a day. Sleep deprivation may lead to a one-third reduction in glucose metabolism and increase in C-reactive protein and interleukin-6 levels. Sleep for longer than 9 hours seems to have similar effects. Onen and colleagues found that men showed hyperalgesia to mechanical stimuli following 40 hours of total sleep deprivation and a robust analgesic effect after selective slow wave sleep recovery.
Sleep deprivation may be dictated by the individual’s lifestyle and habits, but several identifiable sleep conditions may magnify pain symptoms and trigger awakenings. When assessing an individual who has sleep difficulties it is helpful to have a checklist of potential conditions that need to be ruled out or addressed. The first important step in such assessment is to evaluate the patient for sleep disordered breathing. Snoring, awakenings with gasping, palpitations, panic, and dry mouth/sore throat are common symptoms of sleep apnea syndrome. In addition, there are often more subtle symptoms, such as morning headache, anxiety, and poor daytime concentration, that point to potential sleep disordered breathing ( Table 1 ).
| Men | Women | |
|---|---|---|
| Snoring/apneas | ∗∗∗ | ∗ |
| Sleepiness | ∗∗∗ | ∗∗ |
| am Headaches | ∗ | ∗∗∗ |
| Depressive features | ∗ | ∗∗ |
| Apnea frequency | ∗∗ | ∗ |
| Hypopnea frequency | ∗ | ∗∗ |
∗ Qualitative index comparing findings in males and females.
Using a quick screening tool such as the STOP BANG questionnaire is a helpful way to screen many patients.
Snoring (yes/no)
Tired (yes/no)
Observed apneas (yes/no)
Pressure, treatment of blood pressure (yes/no)
Body mass index greater than 35 (yes/no)
Age more than 50 years (yes/no)
Neck circumference greater than 40 cm (yes/no)
Gender male (yes/no).
Answering “Yes” to more than 3 of these questions suggests a high risk of sleep apnea. Patients at risk can then be assessed with a polysomnogram in a sleep laboratory setting or with a home sleep test device, which is especially helpful in severe cases of sleep apnea. If sleep apnea is diagnosed, treatment options include CPAP, oral appliance, weight loss, and surgical intervention. Resultant improvement in sleep continuity and daytime energy level facilitates improved coping with a range of pain symptoms.
The next important step is to assess for abnormal leg movements during sleep and for RLS ( Fig. 2 ).
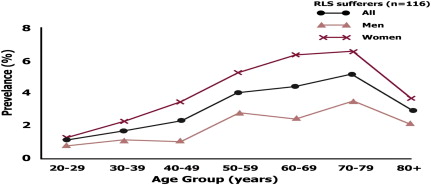
Patients may describe grabbing sensations, sensations that feel creepy-crawly or shocklike, sensations that feel like worms moving, or bubbling in the veins. RLS results in disturbed sleep and sleep onset insomnia and is also associated with a higher risk of depression, anxiety, and somatic pain.
Diagnostic criteria for RLS include:
- 1.
Urge to move: irresistible, involving both legs and may involve arms and trunk
- 2.
Worsening symptoms at rest (body position should not matter)
- 3.
Relief with movement (no symptoms during movement)
- 4.
Worsening in the evening or at night (circadian fluctuation)
RLS may mimic positional discomfort, cramps, positional ischemia, neuropathy, radiculopathy, or hypnic jerks, and should be carefully distinguished from these conditions. RLS is 4 times more likely in first-degree relatives. A polysomnogram is typically unnecessary unless the patient is unable to articulate symptoms.
Although RLS is a condition that the patient is able to report to the provider, periodic limb movements of sleep is a condition diagnosed in the sleep laboratory with recorded periodic episodes of repetitive and highly stereotyped limb movements that occur during sleep and lead to impairment in sleep quality or daytime functioning. Periodic limb movement disorder is defined by the presence of periodic limb movements, electrographic arousals on a sleep study that result in broken sleep, and daytime symptoms of fatigue. Pharmacologic treatments of RLS and periodic limb movement disorder are similar.
Some patients with RLS develop symptoms when ferritin levels decrease to less than 50 ng/mL, and supplementation with iron may improve the symptoms. Other conditions associated with RLS are pregnancy (more commonly the second half), renal insufficiency/failure, neuropathy (Charcot-Marie-Tooth type II only), lumbosacral radiculopathy, anxiety, Parkinson disease, and multiple sclerosis. Dopamine agonists, such as ropinirole and pramipexole, and gamma-aminobutyric acid (GABBA)–ergic (GABA normalizing and increasing) medications are the first approach to the management of restless legs as well periodic limb movements. Long-term use of carbidopa/levodopa may have limited efficacy because of augmentation, which is the process of diminishing efficacy despite dose escalation as frequency of movements, number of duration, body parts affected, and intensity of restlessness worsen with increasing doses. These patients should be cautious in their use of selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors, antihistamines, alcohol, and caffeine because these agents may exacerbate restless legs.
The next step is to assess underlying psychological factors, including pain and mood, which can affect sleep. Patients with pain and/or mood disorders may have excess central autonomic activity triggered by pain as well as peripheral autonomic hyperactivity, including tachycardia, hypertension, mydriasis, vasoconstriction in extremities, and hyperreflexia. Such states of hyperarousal can interfere with the normal process of relaxation that precedes sleep. Because of the difficulty of assessing the direction of cause and effect between insomnia and mood, it is often most effective to manage these conditions by using both pharmacologic and nonpharmacologic approaches.
Pharmacologic approaches to the treatment of insomnia can be split into hypnotic and psychotropic medications that can be used to induce or maintain sleep.
Hypnotics and benzodiazepines carry the risk of dependence and respiratory suppression at high doses, especially if combined with opioid agents. For this reason, careful medication selection is especially critical in patients with pain (because of frequent coadministration of opioids, antiepileptic agents, neuropathic agents, and muscle relaxants). In patients at low risk for addiction, these medications may be a helpful for managing acute insomnia (symptoms lasting less than 30 days). Over-the-counter antihistamines may also be of modest efficacy.
Benzodiazepine receptor agonists are among the most common prescription sleeping aids and include eszopiclone and zaleplon. These agents are highly selective for the alpha-1 subunits of the GABA-a receptors, which facilitates a specific sedative effect. However, adequate time (6–8 hours, depending on the agent) must be allowed to pass between dose administration and the next morning awakening. When blood levels of eszopiclone were checked 8.5 hours after administration, driving impairment equivalent to that of an individual with blood alcohol concentration of 0.5% was observed. Other concerning side effects include daytime sedation, risk of falls (especially in the elderly), confusion, exacerbation of underlying breathing difficulties (most concerning in patients who are also on opioid medications), and short-term memory issues. Sleep walking and sleep eating have been reported with zolpidem specifically but are possible with other hypnotics in the same class.
Benzodiazepines specifically targeting sleep include temazepam, triazolam, and estazolam. Concern remains regarding the effect of benzodiazepines and hypnotics on cognition. A recent case-control study by Billioti de Gage and colleagues found that benzodiazepine use may be associated with higher risk of developing Alzheimer disease (the study tracked individuals more than 66 years of age over 5 years after starting benzodiazepines).
Other medications to consider in the treatment of sleep problems:
- •
Antidepressant medications with sedative properties include tricyclic antidepressants (doxepin, nortriptyline, amitriptyline), the tetracyclic antidepressant mirtazapine, and the selective serotonin reuptake inhibitor trazodone.
- •
In 2014 the FDA approved a novel sleeping aid called suvorexant. It blocks orexin receptors in lateral hypothalamus. Orexin is required for alertness and blocking this pathway may help with sleep induction (the sleep initiation effect is dose related).
- •
Another agent with a unique mechanism of action is ramelteon, which is a melatonin receptor (MT-1 and MT-2) agonist that can be used for sleep initiation difficulties. MT-1 binding inhibits circadian-mediated wake-promoting activity, thereby allowing the brain to turn off. MT-2 receptors are involved in the timing of sleep.
- •
Melatonin (discussed further later) has an important role in sleep initiation because it advances individual circadian clocks by inducing drowsiness earlier in the night, thus allowing earlier sleep onset when used to advance a person’s biological clock.
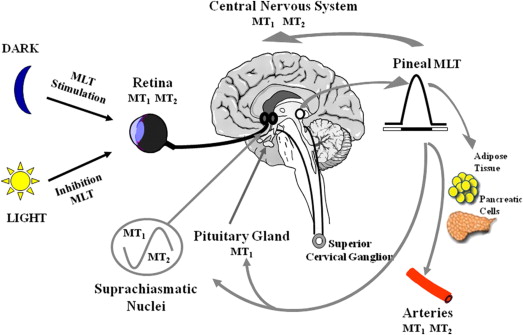
Sleep-wake cycles are intricately linked to multiple environmental and internal stimuli. These circadian rhythms dictate when an individual may become drowsy or wake up. Melatonin is a biogenic amine that is found in humans, animals, and plants. In mammals, melatonin is produced by the pineal gland. Secretion of melatonin increases in darkness and decreases with exposure to light ( Fig. 3 ).