Abstract
Spinal cord injury leads to a range of disabilities, including limitations in locomotor activity, that seriously diminish the patients’ autonomy and quality of life. Electrochemical neuromodulation therapies, robot-assisted rehabilitation and willpower-based training paradigms restored supraspinal control of locomotion in rodent models of severe spinal cord injury. This treatment promoted extensive and ubiquitous remodeling of spared circuits and residual neural pathways. In four chronic paraplegic individuals, electrical neuromodulation of the spinal cord resulted in the immediate recovery of voluntary leg movements, suggesting that the therapeutic concepts developed in rodent models may also apply to humans. Here, we briefly review previous work, summarize current developments, and highlight impediments to translate these interventions into medical practice to improve functional recovery of spinal-cord-injured individuals.
The World Health Organization (WHO) estimates that as many as 500,000 people suffer from a spinal cord injury (SCI) each year. SCI leads to a myriad of detrimental changes in vital physiological and sensorimotor functions, including locomotor impairments that significantly diminish the patients’ quality of life. In most cases of SCI, the spinal neuronal networks that coordinate leg movements are located below the injury. Previous research has shown that even when isolated from supraspinal centers, the spinal cord of rats, cats and humans can still produce motor patterns to stand and walk . However, spinal cord damage interrupts the descending sources of modulation and activation that are essential for the operation of spinal locomotor circuits. Consequently, these networks are in a non-functional state . In the majority of SCIs, however, some nerve fibers still connect the brain and brainstem to the spinal cord below the injury . Yet, more than half of patients with SCI remain bound to a wheelchair for the rest of their lives, and many of the less severe cases continue to suffer from significant motor impairments.
1
Current strategies to improve recovery after SCI
Progress continues in the identification of interventions that show promise in augmenting recovery of motor function after experimental SCIs in rodents . Some of these treatments may be translatable to patients with moderate spinal cord damage , and have entered phase 1 or 2 clinical trials. Nevertheless, clinical evidence for efficacy of any intervention designed to repair the injured human spinal cord is still lacking. Cellular and molecular therapies have shown promise to modify disease or promote regeneration in animal models, but their translation into curative or repair interventions remains a long and uncertain process . In these therapeutic strategies, functional recovery after SCI has been interpreted as the need to promote long-distance regeneration of severed fibers to their original targets. Strategies to promote functional recovery after severe spinal cord damage will undoubtedly incorporate experimental paradigms to regenerate the injured neural pathways. However, a fundamentally new and more immediate approach to improve recovery may instead exploit the intrinsic capacity of sensorimotor circuits within the spinal cord to generate effective postural and locomotor tasks, and the ability of residual descending connections to reorganize with training .
2
Rehabilitative training improves recovery after spinal cord injury
Contrary to nerve-growth promoting interventions, there has been substantial success in translating activity-based rehabilitation from cat models to humans . Improvements of ambulatory function in response to locomotor training in patients with incomplete SCI have been reported in several studies from different laboratories . However, locomotor training has not resulted in successful overground walking in patients with a severe SCI classified as AIS-A, -B, or -C with low motor scores . Failure to promote functional improvement in these patients suggests that although significant control of locomotion can occur at the level of spinal interneuronal networks, the degree of sustainable excitability of these circuits is still compromised. Therefore, a novel and promising strategy to enhance the efficacy of activity-based rehabilitation in severely impaired patients is to develop neuroprosthetic interventions to enable functional states of spinal motor circuits during training .
2
Rehabilitative training improves recovery after spinal cord injury
Contrary to nerve-growth promoting interventions, there has been substantial success in translating activity-based rehabilitation from cat models to humans . Improvements of ambulatory function in response to locomotor training in patients with incomplete SCI have been reported in several studies from different laboratories . However, locomotor training has not resulted in successful overground walking in patients with a severe SCI classified as AIS-A, -B, or -C with low motor scores . Failure to promote functional improvement in these patients suggests that although significant control of locomotion can occur at the level of spinal interneuronal networks, the degree of sustainable excitability of these circuits is still compromised. Therefore, a novel and promising strategy to enhance the efficacy of activity-based rehabilitation in severely impaired patients is to develop neuroprosthetic interventions to enable functional states of spinal motor circuits during training .
3
Neuroprosthetic interventions
Advances in fundamental brain science, neuroengineering, neurosurgery, neuropharmacology, and robotics have opened avenues for alternative and more rapidly applicable solutions to cellular and molecular therapies. This broad field of research—termed neuroprosthetics—taps into spared brain and spinal circuits to restore sensorimotor functions . Regulation of dysfunctional circuits with electrical and/or chemical neuromodulation therapies has broadened the spectrum of treatment options for neurological disorders . For example, dopaminergic replacement therapy and deep brain stimulation of basal ganglia have become common medical practices to alleviate, individually and synergistically, many of the cognitive and motor symptoms of Parkinson’s disease . Over the past decade, much effort has been invested in the development of similar neuromodulation therapies for SCI . A number of neuroprosthetic interventions exploiting chemical and electrical neuromodulation therapies of spinal circuits have been designed to tune the physiological state of spinal circuits to a level sufficient for stepping and standing to occur after SCI .
4
Electrical neuromodulation of spinal circuits
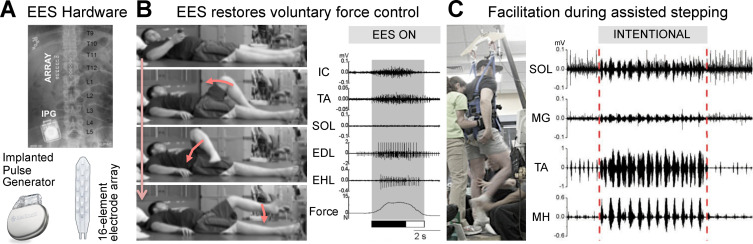
A vast number of studies in various animal models and humans have provided overwhelming evidence that electrical neuromodulation of lumbar segments facilitates motor control after SCI . A recent study has explored the therapeutic potential of electrical neuromodulation in 4 individuals with chronic motor complete paralysis (AIS-A/B) . An electrode array was implanted over the dorsal aspect of lumbar segments ( Fig. 1 A). The array was connected to an implanted pulse generator (IPG) to deliver epidural electrical stimulation, termed EES. Participants followed an 80-session neurorehabilitation program including stand training with EES. At the end of this program, continuous EES enabled full weight-bearing standing for several minutes in all the tested participants. When delivered during manually-assisted stepping, SCI individuals showed the capacity to intentionally assist the trainers in every step, which led to increased muscle activity and occasionally promoted independent steps ( Fig. 1 C). Even more strikingly, with EES all the participants were capable of transforming visual or auditory instructions into a specific motor command to perform graded movements of their paralyzed legs ( Fig. 1 B). The capacity to finely control the force and adjust the level of muscle activity significantly improved with training. These results have two important implications. First, the immediate recovery of motor control capacities during EES indicates that residual descending input can access spinal circuits below the injury when the level of excitability of these neuronal networks is increased with neuromodulation therapies. Second, improvements of supraspinal control of leg movements with training suggest that rehabilitative training enabled by EES promotes neuroplasticity of descending fibers that were presumably spared by the lesion . However, continuous EES alone was not sufficient to raise the functional state of spinal circuits to a level that enables walking overground in severely impaired patients.
5
Chemical neuromodulation of spinal circuits
Brainstem neurons release serotonin (5HT), noradrenaline (NA), and dopamine (DA) within most laminae of lumbosacral segments during locomotion . These monoaminergic inputs primarily operate peri-synaptically through 3-D signal diffusion, which is termed volume neurotransmission. This mode of communication has enabled the design of various monoaminergic replacement therapies. Prescription of L-Dopa has become a common medical practice to compensate for the dopaminergic neuron loss in patients with Parkinson’s disease. Experiments in rodents and cats allowed the identification of various monoaminergic receptors that modulate specific features of spinal locomotor circuits . This knowledge translated into monoaminergic replacement therapies that are able to promote robust locomotion in animal models of incomplete and complete SCI . These chemical neuromodulation therapies interact synergistically with EES. For example, early after a complete SCI, neither chemical nor electrical stimulation is capable of promoting leg movements. However, their combination instantly mediates coordinated weight-bearing locomotion ( Fig. 2 ).
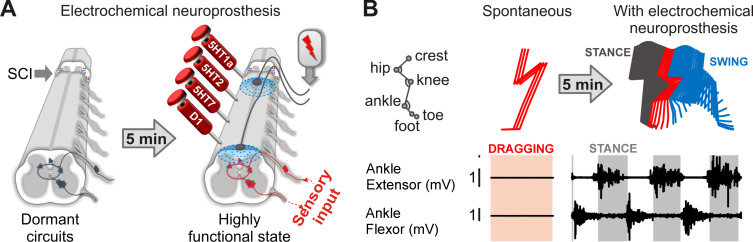
The functional impact of these monoaminergic replacement therapies on the facilitation of locomotion has never been explored in non-human primates and human patients with SCI. Pharmacological experiments in acutely spinalized marmoset monkeys showed that the delivery of serotoninergic and noradrenergic drugs can elicit fictive locomotor states, but the robustness of the motor activity was weak compared to those observed in rodents . However, acute spinalization induces a pronounced and long-lasting depression of spinal circuits in primates compared to lower mammals. Moreover, the presence of sensory information is essential to evaluate the ability of spinal circuits to process information during pharmacologically enabled states, which is absent during fictive locomotion. Finally, the strong synergy between monoaminergic replacement therapies and electrical spinal cord stimulation has never been tested in primates. Future research must dissect circuit-level mechanisms through which activation of monoaminergic receptors enhances the ability of electrical spinal cord stimulation to facilitate locomotion in animal models of SCI. This knowledge may play a key role to design safe and efficacious chemical neuromodulation therapies for humans.
6
Neuroprosthetic rehabilitation augments neuroplasticity and recovery
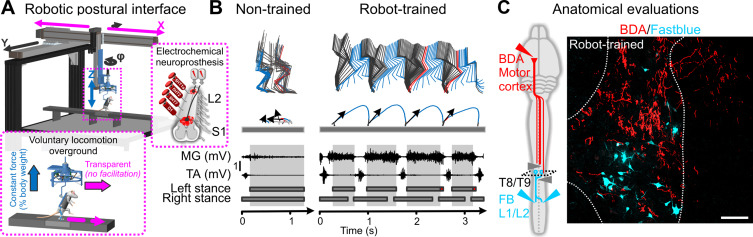
Monoaminergic replacement therapies and EES applied on the dorsal aspect of lumbosacral segments instantly enable locomotion in paralyzed rats. Therefore, this electrochemical neuromodulation therapy provides the opportunity to promote active movement during training in animals with severe SCI. We sought to test the ability of neuroprosthetic rehabilitation to improve functional recovery in a rodent model of SCI leading to complete and permanent paralysis. To enable active rehabilitation, we developed a multidirectional robotic trunk support system that provides adjustable support in the vertical and mediolateral directions . This robot establishes a safe environment that encourages the paralyzed rats to repetitively attempt to engage their legs in order to walk toward a food reward ( Fig. 3 A). This willpower-based training translated into a massive and ubiquitous remodeling of spared spinal circuits below the injury and residual descending connections—an anatomical reorganization that proves the remarkable potential for neuroplasticity even after severe forms of SCI ( Fig. 3 C). Rats with a SCI leading to complete and permanent paralysis are capable of transforming contextual information into specific motor commands to walk overground, and even avoid obstacles and climb staircases ( Fig. 3 B) .