FIGURE 32-1. Artist schematic of spinal cord with motor and sensory (dorsal root ganglion) cell bodies. |
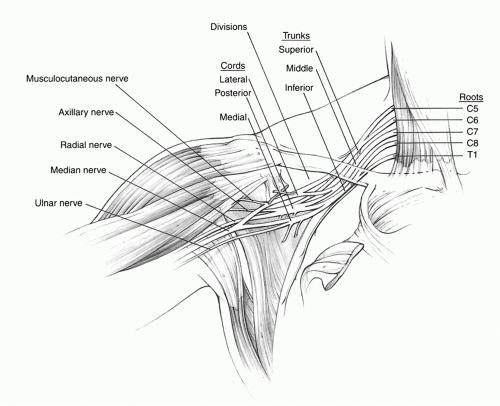
muscle and then becomes the principal motor nerve of the flexor compartment of the arm. The posterior cord divides into the axillary and radial nerves. At the level of the glenohumeral joint, the axillary nerve, along with the posterior humeral circumflex vessels, travels inferior to the subscapularis muscle and across the upper border of the teres major to enter the quadrangular space for innervation of the deltoid and teres minor muscles. Compression of the nerve or artery can occur at the quadrangular space from hypertrophy or anomalies of the bordering muscles. The radial nerve is the largest branch of the brachial plexus and passes inferior to the teres major muscle to enter the posterior arm between the long head of the triceps and humerus via the triangular interval. The medial cord continues as the ulnar nerve and a branch to the median nerve, which contains the vast majority of median motor fibers. The ulnar nerve travels down the arm medial to the brachial artery, pierces the medial intermuscular septum, and enters the cubital tunnel. The median nerve forms anterior to the axillary artery from the union of the medial and lateral and medial cord branches and descends into the arm.
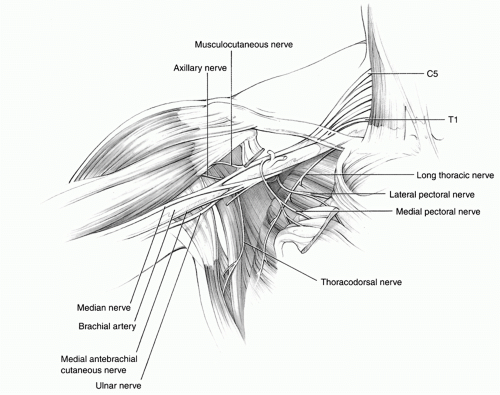
thoracodorsal nerve originates between the upper and lower subscapular nerves, passes behind the axillary artery, and supplies the latissimus dorsi muscle. The medial cord provides one motor and two sensory branches. The medial pectoral nerve traverses and innervates the pectoralis minor muscle and then continues to supply the sternocostal portion of the pectoralis major muscle. The medial brachial and medial antebrachial cutaneous nerves are the only sensory branches to arise directly from the plexus and supply the arm and forearm, respectively. These large sensory nerves can be utilized as a source of nerve grafts in lower trunk injuries, especially in infants as sural nerves have limited length.
These structures include muscles (anterior and middle scalene muscles), skeleton (first rib, cervical ribs, clavicle, and coracoid), and fascia or fibrous bands. The most common sites of compression in thoracic outlet syndrome are at the superior thoracic outlet, the scalene interval or triangle, the costoclavicular space, or the subcoracoid area (Fig. 32-4 Table 32-1).6,149,166,176 This compression can be static or dynamic (i.e., dependent on posture and activity).115
TABLE 32-1 Sites of Compression in Thoracic Outlet Syndrome | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
the coracoid. Excessive arm elevation can lead to a traction or compressive neuropathy along the coracoid process.197
TABLE 32-2 Etiology of Brachial Plexus Injuries | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
suprascapular, or musculocutaneous nerves.4 These injuries are usually (80% of the time) temporary lesions that resolve over 4 to 6 months and are covered in Chapter 33. We have seen permanent injuries affecting various cords and cautious optimism is the rule in plexus injuries following shoulder dislocation.
nutrition and loss of blood supply. Regeneration of the axons occurs at a rate of approximately 1 mm/day or 1 inch/month. This slow regeneration delays recovery and means that distal nerve injuries have a better prognosis because the extent of Wallerian degeneration is decreased and the proximity to the motor endplates is increased. Prolonged denervation (greater than 18 to 24 months) results in irreversible motor endplate degradation and muscle fibrosis. In contrast, the encapsulated sensory receptors retain their capacity for reinnervation for many years. These factors portend the overall prognosis for axonotmesis as variable and guarded, which is vital information to relay to the patient.
Table 32-3 Patterns of Brachial Plexus Injuries | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Table 32-4 Seddon’s Classification of Nerve Injury | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||
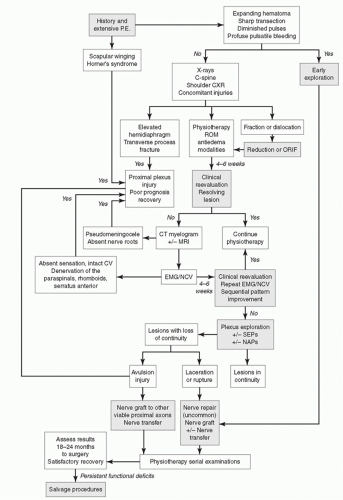
plexus is performed to assess for the presence or absence of a Tinel’s sign. A positive Tinel’s sign is indicative of a postganglionic injury (e.g., rupture), whereas this sign will be absent in a preganglionic lesion (e.g., avulsion) because the link to the spinal cord and brain has been disrupted.
TABLE 32-5 Brachial Plexus Examination Sheet | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 32-6 Indicators of Avulsion Injuries and Poor Prognosis for Recovery | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
 FIGURE 32-7. A 30-year-old woman involved in a motor vehicle accident with avulsions of C8, T1, and ruptures of C5, C6, C7. Persistent subtle left Horner’s syndrome years after injury. |
TABLE 32-7 Practical Examination for Brachial Plexus Injury Pattern | ||||||||
|---|---|---|---|---|---|---|---|---|
|
TABLE 32-8 Radiographic Findings in Injuries to the Brachial Plexus | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
 FIGURE 32-8. Artist schematic of pseudomeningocele associated with avulsion injuries of the brachial plexus. |
however, is the inability to quantify electrical recordings with extent of recovery. In other words, return of electrical activity may not correlate with return of active motion, especially in birth palsies.123,212
electrode to the central nervous system and can detect lesions within the sensory system. This technique can be employed to assess conduction across the brachial plexus to define irreparable nerve root avulsions. The absence of SEPs recorded over the spinal cord or somatosensory cortex on nerve root stimulation indicates a dorsal root avulsion and is a contraindication for nerve grafting.111,142 MEPs are recorded from muscles following stimulation of the motor cortex. Recordings are obtained as neurogenic potentials in the brachial plexus or as myogenic potentials from the muscle. The absence of MEPs via stimulation of the motor cortex indicates a ventral root avulsion and is a contraindication for nerve grafting.
is often frustrating, as the vascular repair cannot be adequately mobilized for fear of disruption. Once the injured nerve segments are identified, the treatment depends upon the status of the damaged segments. Sharp transections can be managed by direct repair or graft interposition. Primary neurorrhaphy requires approximation of the cut ends without excessive tension. 51,136,185 However, primary neurorrhaphy is not recommended for traction or gunshot injuries. Instead, delayed management is preferred to allow demarcation of the zone of injury and extent of nerve damage. Fortunately, most penetrating trauma by missile preserves nerve continuity.18,100,152
 FIGURE 32-11. A 20-year-old with left global plexus injury secondary to motor vehicle accident and chronic refractory pain. (Courtesy of Shriners Hospital for Children, Philadelphia.) |
Table 32-9 Hospital for Sick Children Active Movement Scale (AMS) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
ligated. Next, the phrenic nerve is identified as it travels on the anterior scalene muscle just anterior to the posterior triangle. The phrenic nerve must be protected throughout the dissection (Fig. 32-12C) but the C5 contribution can be sacrificed to gain more proximal root exposure if necessary. Our next step is to isolate the upper trunk and the C5 and C6 roots between the anterior and middle scalene muscles and look for neuroma (Fig. 32-12D). The middle trunk and C7 is inferior and posterior to the upper trunk, whereas the lower trunk is deep to the subclavian vessels and the clavicle, both of which can impede inferior dissection. Osteotomy of the clavicle and/or infraclavicular exposure may be necessary to enhance lower trunk exposure, especially if a clavicular malunion is present.
51,101,141,143 Our preference is biologic adhesives to decrease operative time.63 The laboratory and clinical data demonstrate that glue is equal to, if not superior to, conventional suture methods with regard to the amount of fibrosis, axonal regeneration, and alignment of fascicles. This is due to the fact that the glue acts a sealant and not a barrier to nerve regeneration. Glue does exhibit less tensile strength compared to suture and should not be used alone when the repair is taut.59,153,201
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree