Standard surgical approaches for Perthes disease consist of nonoperative physical treatments or surgical treatment. Several investigators have been working on a better understanding of the pathophysiology and pathobiology of Perthes disease. Most of the focus has been on antiresorptive treatments. Other treatment avenues, however, include controlling the inflammatory phase of Perthes disease, increasing revascularization of necrotic bone, and anabolic strategies to increase bone formation. This article presents a current pathophysiological model of Perthes disease, reviews experimental strategies in pharmaceutical treatments, and suggests future areas for research.
Current nonoperative treatment for Perthes disease, Scottish Rite orthosis, does not seem to affect outcome. The effects of surgical treatments, such as a femoral varus osteotomy and the Salter’s innominate osteotomy, also had moderate effects in the multicenter prospective study by Herring and colleagues. For example, in lateral pillar B hips greater than age 8 years where the effect of surgery was greatest, the rate of spherical hips increased from 44% to 73%. This means that in this favorable scenario, while 29% of the patients operated gained a spherical hip that would not have occurred in the natural evolution of the disease, 71% of patients did not benefit, as 44% would have had a spherical head anyway, and 27% did not achieve sphericity even with surgery. While the effects of surgery are definite, they are by no means universally positive.
Pathology of Perthes disease
Perthes disease can be defined as idiopathic avascular necrosis of the femoral head in childhood. The initiating insult triggering the onset of Perthes disease remains unknown, making a true description of the pathology difficult. However, a great deal more is known about Perthes disease pathogenesis, much of which has been gained by correlating the few true histologic specimens with radiographs, and also by examining animal models of Perthes disease. Although trauma may play a part in Perthes disease, it is unlikely to be the only factor, as traumatic avascular necrosis often behaves differently from Perthes disease. In truth, the initiating factor remains unknown.
However, what is clear is that the femoral head blood supply is very dependent on the ascending branches of the lateral femoral circumflex, in turn a branch of profunda femoris. A condition very similar but not identical to Perthes can be created in piglets by tying off these and other ascending vessels, along with the repetitive loading of the hip joint. In Perthes disease, it is well known that the bone age is delayed, an average of 2 years in girls and 1 year in boys. This means the amount of cartilage present in the developing femoral head is larger and the ossific nucleus smaller. In one theory, these vessels have to traverse a larger-than-normal cartilaginous anlage to get to the epiphysis and are thus vulnerable to mechanical compression. A particular strain of rat called the SHR rat also has a delayed bone age and the onset of a Perthes-like condition occurs in 50% of male rats that stand on their hind limbs to feed. When these rats are prevented from weight bearing, avascular necrosis does not occur.
The few human specimens available show an enlarged cartilaginous anlage, fibrocartilaginous and fibrovascular proliferation, and a disorganization of the growth plate. Depending where the biopsy is taken, areas of granulation tissue or endochondral ossification can be seen. Some of the new bone that has managed to form can be seen to be reinfarcted.
The stages of Perthes disease are well accepted: (1) initial, (2) fragmentation, (3) reossification, and (4) healed. In the initial phase, the ossific nucleus is not growing due to the avascular necrosis, but the cartilaginous anlage continues to enlarge, receiving nutrition from the synovial fluid. Next the femoral head starts to collapse. The pathology here (at least from animal models) is that osteoclasts are removing the dead bone. Resorptive phase may be a better term for this stage than fragmentation. There is an uncoupling of the normal bone remodeling mechanism whereby bone resorption is followed by bone formation. This gives the appearance on radiographs of a central sequestrum that continues to mineralize and is dense, and surrounding lytic areas yet to ossify. Eventually the neo-cartilage in the femoral head undergoes endochondral ossification with the invasion of blood vessels followed by osteoblasts. Eventually all the dead bone is removed and replaced with new bone. This bone is then remodeled into lamellar bone, and the process is complete. However, it is important to note that the lag between resorption and formation leave the largely cartilaginous head very soft and subject to the deformation of the loads placed on it.
Finally, the circulation re-establishes itself, and ossification proceeds from lateral to medial and posterior to anterior. Shape changes continue to occur during the remodeling phase; these can be either beneficial or harmful.
Magnetic resonance imaging (MRI) studies of Perthes disease attest to significant inflammatory changes in the synovium and ligamentum teres, contributing to subluxation.
Hypotheses generated from radiological and pathophysiological findings
Inflammatory Changes
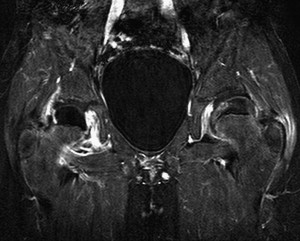
The inflammatory changes in Perthes disease are most marked during the fragmentation phase ( Fig. 1 ). As this phase is when bone resorption and bone formation are uncoupled, it may be that inflammatory mediators are inhibiting bone formation. Such phenomena are well known to disturb the anabolic/catabolic balance in rheumatoid arthritis. Osteoblasts cannot differentiate from mesenchymal stem cells in the presence of high levels cytokines such as tumor necrosis factor (TNF)-α and interleukin (I1-β). If the phathophysiological mechanisms are similar, controlling inflammation may play some role in encouraging new bone formation as well as decreasing bone resorption.
Bone Resorption
While eventual resorption of the necrotic sequestrum would be necessary to restore the normal mechanical properties of the femoral head, rapid resorption leads to collapse. A great deal of research has been performed looking at the role of bisphosphonates in models of Perthes disease. In rodent traumatic models, femoral head structure can be almost completely preserved with zoledronic acid or alendronate treatment. In the SHR model, zoledronic acid can improve femoral head shape.
In the piglet model of ischemic necrosis where the femoral head vessels are disrupted by a ligature, systemic ibandronate significantly improves the maintenance of femoral head shape. However, the biodistribution of bisphosphonate to the necrotic bone is initially poor, favoring local delivery. Treatment with an inhibitor of RANKL, osteoprotegerin (OPG), was systemically effective but also had major effects on growth plate remodelling.
In small animal models, the return of bone formation is rapid with bisphosphonate therapy. However, this finding is not observed in the piglet model or in patients with Perthes disease. This suggests that while anticatabolic strategies are likely to be necessary to preserve femoral head shape, stimulation of bone formation is likely to be also desirable.
Bone Formation
An anabolic strategy would increase revascularization and couple this with increased bone formation. Bone morphogenic proteins (BMPs) and bisphosphonates are known to be synergistic in bone repair. In the piglet model, this experimental strategy used BMP-2 injection at the same time as local ibandronate infusion as a single intraosseous injection. Compared with the control group, the combined therapy group had a significant decrease in femoral head deformity and the osteoclast number and a significant increase in the trabecular bone volume ( Fig. 2 ). The combined treatment group also had a significantly higher osteoblast surface compared with the local ibandronate treatment group, suggesting an increased bone formation as a result of adding BMP-2. One cautionary finding was the presence of heterotopic ossification in the hip joint capsule. This was postulated to be due to a leakage of BMP-2 into the joint during and following the local injection procedure. Further studies underway to determine whether the heterotopic ossification can be prevented by lowering the dose of BMP-2 and changing the delivery technique.


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree





