Intervertebral disks are biologically regulated by the maintenance of a balance between the anabolic and catabolic activities of disk cells. Therapeutic agents, initially evaluated using in vitro studies on disk cells and explants, have been used as intradiscal injections in preclinical settings to test in vivo efficacy. These include anabolic growth factors, other biostimulatory agents, and antagonistic agents against matrix-degrading enzymes and cytokines. Additional work is needed to identify patient populations, using methods such as MRI, and to better understand the mechanism of healing. Clinical trials are underway for a few of these agents and other promising candidates are on the horizon.
Intervertebral disk degeneration and homeostasis of the extracellular matrix
Intervertebral disk (IVD) degeneration is one of the major causes of low back pain and lumbar disk herniation. Biologically, the cells in the disk actively regulate the homeostasis of the IVD extracellular matrix (ECM) by maintaining a balance between anabolism and catabolism. The modulation of disk cell metabolism involves a variety of molecules (eg, cytokines, enzymes, enzyme inhibitors, and growth factors) that act in a paracrine and/or autocrine fashion. The degeneration of an IVD may result from the loss of steady state metabolism maintained in the normal disks, due to an imbalance between the anabolic and catabolic processes. The proteoglycan (PG) content of the ECM and the rate of PG synthesis decreased markedly with age and degeneration, similar to the case in articular cartilage. The anabolic regulators include polypeptide growth factors, such as insulinlike growth factor 1 (IGF-1), transforming growth factor β (TGF-β), and the bone morphogenetic proteins (BMPs). Catabolic regulators include many cytokines, notably interleukin 1 (IL-1) and tumor necrosis factor (TNF)-α), which influence the synthesis of matrix-degrading enzymes, such as the matrix metalloproteinases (MMPs). Alterations in both anabolic and catabolic processes are thought to play key roles in the onset and progression of IVD degeneration; the biochemical processes that regulate these changes are important to understand for the development of effective treatments of disk degeneration.
Catabolic mediators and enzymes in disk degeneration
Under pathologic conditions, including IVD degeneration, physical injury (eg, puncture or stab wounds), and abnormal mechanical loading, the IVD expresses cytokines and proteinases. Although macrophages that infiltrate herniated tissue or granulation tissue seem to be the major source of these cytokines, recent studies indicate that IVD cells may also synthesize these molecules in an autocrine fashion. In particular, increases in both protein and mRNA levels of IL-1α and its major regulator, TNF-α, have been observed in degenerated or herniated IVD tissues and both are spontaneously expressed by these tissues in culture.
The catabolic processes induced by cytokines are mediated by various enzymes, such as the MMPs or the aggrecanases. IL-1 has been shown to stimulate matrix degradation as well as to inhibit the synthesis of ECM macromolecules with minimum effect on cell proliferation. IL-1 induced the production of collagenase, cyclooxygenase-2 (COX-2 ), prostaglandin E2, nitric oxide, MMP-1, total and active MMP-3, MMP-13, the aggrecanase A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS)-4), IL-6, and monocyte chemoattractant protein-1. Recently, IL-1β was shown to increase the sensitivity of IVD cells to shear stress, suggesting that this cytokine is involved in the acceleration of degradation processes in IVDs subjected to biomechanical stress. Although IL-1 affects both synthetic and degradative processes, at lower concentrations, IL-1 was much more effective in inhibiting the synthesis of aggrecan than in stimulating its degradation in articular cartilage. An epidemiologic study has shown that IL-1 gene cluster polymorphisms significantly increased the risk of disk degeneration; this has shed more light on the possible involvement of IL-1 in IVD degeneration.
The effects of the inhibition of catabolic cytokines have been evaluated in several studies. IL-1 receptor antagonist (IL-1ra), applied in vitro to degenerated and herniated human disk tissues, reduced the expression of MMP-3. IL-1ra pretreatment of nucleus pulposus (NP) cells from moderately degenerate human disks reduced the expression of ADAMTS-4 and MMP-3 in subsequent treatment with IL-1. In another human cell culture study, the addition of IL-1ra and soluble TNF receptor significantly up-regulated PG synthesis, suggesting that IL-1 and TNF suppress PG de novo synthesis. IL-1ra and other agents may benefit from administration of slow-release agents, such as IL-1ra mixed with thermally responsive elastin-like polypeptide (ELP) or with gelatin hydrogel. In addition to IL-1, TNF-α has gained attention in association with disk herniation. TNF-α inhibition, using a monoclonal antibody in herniated human disk explants, showed suppression of MMP-3 levels ; other anti-TNF agents are beginning to be applied to reduce pain in patients with sciatica and discogenic pain. Other anticytokine therapeutics include the p38 mitogen-activated protein kinase (MAPK) inhibitor, which hindered the catabolic effects of IL-1, as well as nuclear factor κB decoy, which reduced pain in a rat lumbar disk herniation model.
Catabolic mediators and enzymes in disk degeneration
Under pathologic conditions, including IVD degeneration, physical injury (eg, puncture or stab wounds), and abnormal mechanical loading, the IVD expresses cytokines and proteinases. Although macrophages that infiltrate herniated tissue or granulation tissue seem to be the major source of these cytokines, recent studies indicate that IVD cells may also synthesize these molecules in an autocrine fashion. In particular, increases in both protein and mRNA levels of IL-1α and its major regulator, TNF-α, have been observed in degenerated or herniated IVD tissues and both are spontaneously expressed by these tissues in culture.
The catabolic processes induced by cytokines are mediated by various enzymes, such as the MMPs or the aggrecanases. IL-1 has been shown to stimulate matrix degradation as well as to inhibit the synthesis of ECM macromolecules with minimum effect on cell proliferation. IL-1 induced the production of collagenase, cyclooxygenase-2 (COX-2 ), prostaglandin E2, nitric oxide, MMP-1, total and active MMP-3, MMP-13, the aggrecanase A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS)-4), IL-6, and monocyte chemoattractant protein-1. Recently, IL-1β was shown to increase the sensitivity of IVD cells to shear stress, suggesting that this cytokine is involved in the acceleration of degradation processes in IVDs subjected to biomechanical stress. Although IL-1 affects both synthetic and degradative processes, at lower concentrations, IL-1 was much more effective in inhibiting the synthesis of aggrecan than in stimulating its degradation in articular cartilage. An epidemiologic study has shown that IL-1 gene cluster polymorphisms significantly increased the risk of disk degeneration; this has shed more light on the possible involvement of IL-1 in IVD degeneration.
The effects of the inhibition of catabolic cytokines have been evaluated in several studies. IL-1 receptor antagonist (IL-1ra), applied in vitro to degenerated and herniated human disk tissues, reduced the expression of MMP-3. IL-1ra pretreatment of nucleus pulposus (NP) cells from moderately degenerate human disks reduced the expression of ADAMTS-4 and MMP-3 in subsequent treatment with IL-1. In another human cell culture study, the addition of IL-1ra and soluble TNF receptor significantly up-regulated PG synthesis, suggesting that IL-1 and TNF suppress PG de novo synthesis. IL-1ra and other agents may benefit from administration of slow-release agents, such as IL-1ra mixed with thermally responsive elastin-like polypeptide (ELP) or with gelatin hydrogel. In addition to IL-1, TNF-α has gained attention in association with disk herniation. TNF-α inhibition, using a monoclonal antibody in herniated human disk explants, showed suppression of MMP-3 levels ; other anti-TNF agents are beginning to be applied to reduce pain in patients with sciatica and discogenic pain. Other anticytokine therapeutics include the p38 mitogen-activated protein kinase (MAPK) inhibitor, which hindered the catabolic effects of IL-1, as well as nuclear factor κB decoy, which reduced pain in a rat lumbar disk herniation model.
Anabolic effects of cytokines and growth factors on IVD cells
A variety of growth factors and cytokines ( Table 1 ) can alter IVD homeostasis and stimulate ECM synthesis by shifting cellular metabolism to a more anabolic state. The effects of TGF-β on PG synthesis and cell proliferation were described early in the literature. Similar effects of IGF-1 have also been reported. IGF-1 and platelet-derived growth factor (PDGF) were also shown to reduce the percentage of apoptotic anulus fibrosus (AF) cells induced by serum depletion in culture.
| Agent | Target | Dose | Effect | Author |
|---|---|---|---|---|
| TGF-β | Mature canine IVD | 1 ng/mL | PG synthesis increased up to 5×; higher in NP than AF | Thompson et al, 1991 |
| IGF-1 | Mature canine IVD | 20 ng/mL | PG synthesis marginally increased in NP | Thompson et al, 1991 |
| TGF-β | Human anulus cells, 3-D culture | 0.25–5 ng/mL | Cell proliferation and PG synthesis increased; reduced apoptosis with serum depletion | Gruber et al, 1997 |
| IGF-1 | Young and old bovine NP cells | 0–1000 ng/mL | Cell proliferation and matrix synthesis stimulated; more IGF-1 receptors | Osada et al, 1996 |
| OP-1 | Young rabbit NP and AF cells; alginate beads | 0, 100, 200 ng/mL | Increased PG and collagen production and content | Masuda et al, 2003 |
| BMP-2 | Rat IVD cells monolayer | 0–1000 ng/mL | Increased cell number, GAG, expression for collagen, aggrecan at higher doses | Yoon et al, 2003 |
| OP-1 | Human NP and AF cells, alginate beads | 0, 100, 200 ng/mL | Maintained cell density, increased PG synthesis and accumulation | Imai et al, 2007 |
| OP-1 | Rabbit IVD cells; alginate beads: IL-1α pre-exposure | 200 ng/mL | IL-1α decreased PG and collagen; reversed and exceeded with OP-1 | Takegami et al, 2002 |
| OP-1 | Rabbit IVD cells; alginate bead: C-ABC pre-exposure | 200 ng/mL | OP-1 up-regulated PG synthesis; greater effect after C-ABC pre-Tx than control | Takegami et al, 2005 |
| BMP-2 | Human IVD cells | 300, 1500 ng/mL | Increased PG synthesis, expression of aggrecan, collagen types I and II; no bone formation | Kim et al, 2003 |
| rhBMP-2 & 12 | Human IVD cells in monolayer | 25–300 ng/mL | PG, collagen synthesis increased in NP cells; minimal effect on AF cells | Gilbertson et al, 2008 |
| GDF-5 | Bovine IVD cells; alginate bead | 100, 200 ng/mL | Increased DNA and PG content; at higher dose, PG and collagen synthesis increased | Chujo et al, 2006 |
| PRP | Porcine IVD cells; alginate bead | 10% PRP | Mild increase in cell proliferation; marked increase in PG and collagen synthesis and PG accumulation | Akeda et al, 2006 |
| TGF-β1 and PRP | Human NP cells | 1 ng/mL TGF-β1 in PRP | NP cell proliferation and aggregation; increase in mRNA of SOX-9, collagen type II, aggrecan | Chen et al, 2006 |
| Ad-TIMP-1, Ad-BMP-2 | IVD cells from human degenerated IVD | 50–150 MOI | 2000 pg/mL production of TIMP w/100 MOI at day 4; PG synthesis increased with both Ad-TIMP-1 and Ad-BMP-2 | Wallach et al, 2003 |
| Dexa-methasone | Human disk herniation tissue explants | 0.01 mM | Decreased MMP-1 and -3 levels | Genevay et al, 2009 |
| IL-1ra | Human disk herniation tissue explants | 100 ng/mL | Decreased MMP-3 levels | Genevay et al, 2009 |
| IL-1ra | Human normal and degenerated disk tissues in situ with IL-1β treatment | 100 ng/mL | IL-1ra reduced cytokine levels (MMP-3, -7, -13) and matrix degradation in all tissue types | Le Maitre et al, 2007 |
| IL-1ra/ELP | Human IVD cells (grade 2–3); alginate beads: IL-1ra pre-Tx, then IL-1β insult | 4–8000 pM | Reduced ADAMTS-4, MMP-3 transcription | Shamji et al, 2007 |
| p38 MAPK inhibitor (SB 202,190) | Rabbit NP cells pre-Tx with IL-1 | 1 μM | Decreased message for collagen, aggrecan, IGF-1; increased message for iNOS, COX-2, MMP-3, IL-6 | Studer et al, 2008 |
| TNF inhibitor mAb | Human IVD herniation tissue explants | 10 μg/mL | Decreased MMP-3 levels | Fujita et al, 1993 |
Osteogenic protein-1 (OP-1), which is a member of the BMP family, up-regulates the PG metabolism of IVD cells. OP-1 strongly stimulated the production and formation of the ECM by rabbit IVD cells ; similar effects were found using human IVD cells. OP-1 also replenished PGs and collagens after depletion of the ECM after exposure of IVD cells to IL-1 or chondroitinase ABC (C-ABC).
Growth and differentiation factor-5 (GDF-5), originally found to be a factor responsible for skeletal alterations in brachypodism mice, is another member of the BMP family that has anabolic effects on IVD cells. GDF-5 stimulated PG and collagen type II expression by mouse IVD cells. Recombinant human GDF-5 (rhGDF-5) enhanced cell proliferation and matrix synthesis and accumulation by cells from bovine NP and, to lesser extent, AF cells.
Because biologic molecules, prepared in an autologous fashion, avoid certain regulatory complications, they may be useful clinically. Autologous IL-1ra has been used, along with IGF-1 and PDGF proteins, to reduce apoptosis of disk cells and their production of IL-1 and IL-6. Platelet-rich plasma (PRP) can be produced by centrifugal separation of a patient’s own blood in the operating room; it contains multiple growth factors concentrated at high levels. In vitro, PRP stimulated cell proliferation and matrix synthesis, as shown using porcine disk cells. PRP induced cell proliferation and differentiation and facilitated NP-like tissue formation by human disk cells.
IntradisCal therapeutics: animal studies
To study the ability of growth factors and other therapeutic agents to stimulate repair in vivo ( Table 2 ), small animal models, in which the degree of degeneration can be controlled, are useful. Rabbit lumbar and rat caudal disks have been used extensively, although other animal models exist. Both physical injury, such as stab wound or controlled needle puncture, and chemical degradation approaches have been used.
| Agent | Species | Site | Model | Dose per Disk | Effect | Author |
|---|---|---|---|---|---|---|
| IGF-1 | Rat | Tail | Static compression | 8 ng/8 μL | Clustering of inner anulus cells after single injection | Walsh et al, 2004 |
| GDF-5 | Rat | Tail | Static compression | 8 ng/8 μL | Clustering of cells, increase in disk height (single injection) | Walsh et al, 2004 |
| TFG-β | Rat | Tail | Static compression | 1.6 ng/8 μL | Proliferation of cells (multiple injections) | Walsh et al, 2004 |
| bFGF | Rat | Tail | Static compression | 8 ng/8 μL | No response | Walsh et al, 2004 |
| OP-1 | Rabbit | Lumbar | None (normal) | 2 μg/10 μL | Increased disk height and PG content in NP | An et al, 2005 |
| OP-1 | Rabbit | Lumbar | C-ABC: Co-injection | 100 μg/10 μL | Increased disk height and PG content in NP | Imai et al, 2003 |
| OP-1 | Rabbit | Lumbar | Needle puncture: Tx 4 wk later | 100 μg/10 μL | Increased disk height and PG content in NP and AF, improvement of MRI and histology grades | Masuda et al, 2006 |
| OP-1 | Rabbit | Lumbar | Needle puncture | 100 μg/10 μL | Increased disk height and viscoelastic properties | Miyamoto et al, 2006 |
| OP-1 | Rabbit | Lumbar | C-ABC: Tx 4 wk later | 100 μg/10 μL | Increased disk height, PG content in NP and AF | Imai et al, 2007 |
| GDF-5 | Rabbit | Lumbar | Needle puncture: Tx 4 wk later | 1, 100 ng/10 μL; 1, 100 μg/10 μL | Increased disk height, improvement of MRI and histology grades | Chujo et al, 2006 |
| GDF-5 | Rabbit | Lumbar | Thrombin-degraded: Tx 4 wk later | 10 μg/10 μL | Increased disk height, improved T1rho and T2 values; decreased ADAMTS-4, -5, COX-9 expression | Bae et al, 2009 |
| BMP-2 | Rabbit | Lumbar | Anular stab (5 × 7 mm) | 100 μg/100 μL | More degeneration, vascularity and fibroblast | Huang et al, 2007 |
| PRP | Rabbit | Lumbar | Nucleotomy, immediate Tx | PRP + GHM (20 μL) or PRP (5 μL) + PBS (15 μL) | PRP + GHM group had less degeneration and increased PG; PRP + PBS group showed no differences | Nagae et al, 2007 |
| PRP | Rabbit | Lumbar | Nucleotomy, immediate Tx | 20 μL of PRP + GHM, PBS + GHM, PRP + PBS; puncture-only | PRP + GHM had greater disk height, water content, mRNA for PG core protein and collagen type II collagen; fewer apoptotic cells in NP | Sawamura et al, 2009 |
| BMP-17 | Sheep | Lumbar | Anular stab (3 × 6 mm), immediate Tx | 300 μg/70 μL | BMP-17 maintained disk height, MRI and histology scores, NP cell density; increased PG and collagen synthesis | Wei et al, 2009 |
| ADAMTS-5 siRNA | Rabbit | Lumbar | Needle puncture: Tx 4 wk later | 10 μg/10 μL | Improved MRI and histology scores | Seki et al, 2009 |
The in vivo efficacy of OP-1 injection has been evaluated in several animal studies. In adolescent rabbits, an injection of recombinant human OP-1, but not the lactose vehicle, reversed the reduction in disk height and worsened the magnetic resonance imaging (MRI) grade caused by an anular needle puncture. In another study with the same experimental design, the injection of OP-1 restored dynamic viscoelastic biomechanical properties, such as elastic and viscous moduli, of puncture-degenerated IVDs. OP-1 treatment was also effective in restoring disks that have been chemically degraded. C-ABC has been used as an alternative to chymopapain for chemonucleolysis as well as an animal model of disk degeneration in the rat tail and goat disk. When OP-1 or vehicle was injected into rabbit disks degraded with C-ABC for 4 weeks, the disk height initially decreased (approximately 34%), then recovered and gradually approached the level of the normal control.
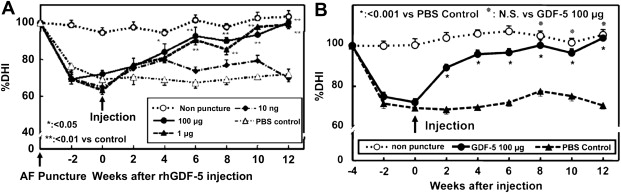
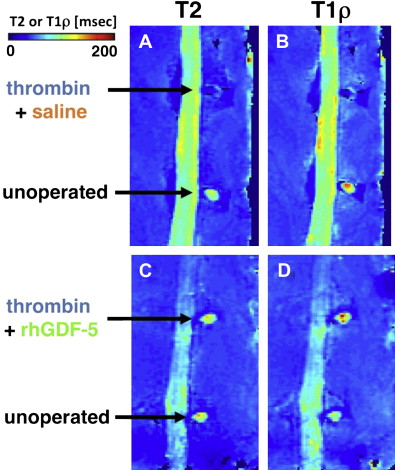
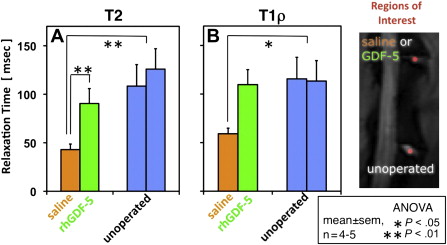
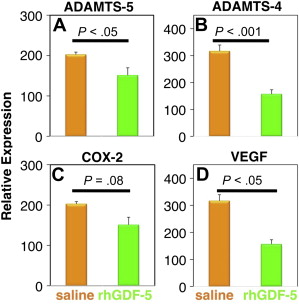
rhGDF-5 is another promising growth factor whose efficacy has been evaluated in animal models. The efficacy of injecting a single dose of GDF-5 has been reported in the mouse caudal disk with degeneration induced by static compression. In that study, GDF-5 induced an increase in disk height and the expansion of the inner AF fibrochondrocytic population into the NP. In a needle-puncture model of disk degeneration (4 weeks) in adolescent (5–6 months old) rabbits, a single injection of rhGDF-5 restored disk height ( Fig. 1 A) and improved MRI and histologic grading scores within approximately 6 weeks. There are concerns that aged or degenerated disks might have lower cellularity, especially in the number of notochordal cells often found in normal adolescent rabbit disks, and might respond poorly to growth factor treatment. Despite this, a study using 2-year-old rabbits showed that rhGDF-5 effectively recovered disk height (see Fig. 1 B) as well as MRI and histologic grades after a 12-week observation period. Biomechanical analyses indicated that the viscous and elastic moduli of the IVDs in the rhGDF-5-injected disks were significantly higher than those in the phosphate-buffered saline (PBS)-injected disks. Additionally, rhGDF-5 has shown efficacy in rabbit disks that have been degraded using thrombin, a serine proteinase that results in cleavage of PGs and decreased disk height. In this study, disks of adolescent rabbits were injected with thrombin (100 U/10 μL) and the rabbits were maintained for 4 weeks. A single injection of rhGDF-5 (10 μg/10 μL) or saline was then given, and endpoint measures (MRI and gene expression) were determined 12 weeks later. Quantitative MRI was used to evaluate T2 and T1rho magnetic resonance properties. T2 ( Fig. 2 A, C) and T1rho (see Fig. 2 B, D) MRI maps showed maintenance of NP morphology and MRI values in rhGDF-5–treated disks. Both T2 ( Fig. 3 A) and T1rho (see Fig. 3 B) values of the NP were higher in the rhGDF-5 group, approaching those of unoperated controls. Treatment with rhGDF-5 also reduced the level of expression for ADAMTS-5 ( Fig. 4 A), ADAMTS-4 (see Fig. 4 B), COX-2 (see Fig. 4 C), and vascular endothelial growth factor (VEGF) (see Fig. 4 D), molecules related to PG degradation, pain, and neovascularization seen in degenerated IVDs.
PRP is another agent that has been evaluated in animal models. Using a nucleotomy rabbit model, the effects of allograft PRP with or without gelatin hydrogel microspheres (to provide slow release and mechanical support) and PBS-only injectates were evaluated. The PRP with microspheres group had markedly suppressed degeneration, compared with the PBS-only and PRP-only groups. A recent study by the same group reported additional findings that microspheres alone without PRP did not have therapeutic value, and that the PRP with microsphere group benefited from increased disk height, water content, expression of PG core protein and collagen II, and fewer apoptotic cells in the NP. Although the use of a sustained delivery system has been effective in nucleotomy models in a less severe anular puncture model, an injection of PRP alone was sufficient to induce restoration of disk height and T2 MRI values. The use of fibrin glue alone had a positive effect in a porcine nucleotomy model, where the fibrin glue resulted in the suppression of IL-6 and TNF expression, while restoring mechanical properties and glycosaminoglycan (GAG) content.
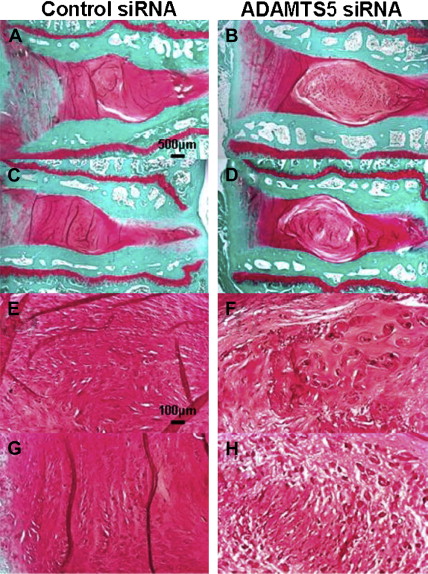
A recent study has developed and evaluated the efficacy of an inhibitor for ADAMTS-5 expression using small interference RNA (siRNA). First, the ability of siRNA to reduce ADAMTS-5 expression was determined using rabbit NP cells in vitro. Adolescent rabbit disks, punctured in the anulus to induce degeneration, were injected with either ADAMTS-5 or control siRNA. After 8 weeks, the disks receiving ADAMTS-5 siRNA had markedly better MRI and histology results ( Fig. 5 ). The control group exhibited complete loss of NP tissues (see Fig. 5 A, C) that had been replaced by a fibrocartilaginous tissue (see Fig. 5 E, G). In contrast, the ADAMTS siRNA group had maintained disk structure (see Fig. 5 B, D), including a clearly distinguishable NP. Both MRI grade ( Fig. 6 A) and overall histology grade (see Fig. 6 B) were significantly better for the ADAMTS siRNA group. The inhibition of degradative enzymes and catabolic cytokines provides a complimentary approach to treat degenerated disks at different levels of the modulation mechanism.